Content
- 1 What Is N-Methylimidazolium Hydrogen Sulfate?
- 2 Chemical Identity and Structural Characteristics
- 3 Key Physical and Chemical Properties
- 4 Role as an Acid Catalyst in Organic Synthesis
- 5 Biomass Processing and Cellulose Dissolution
- 6 Biodiesel Synthesis and Esterification Catalysis
- 7 Electrochemical Applications and Proton Conduction
- 8 Handling, Safety, and Environmental Considerations
- 9 Summary of Principal Uses
What Is N-Methylimidazolium Hydrogen Sulfate?
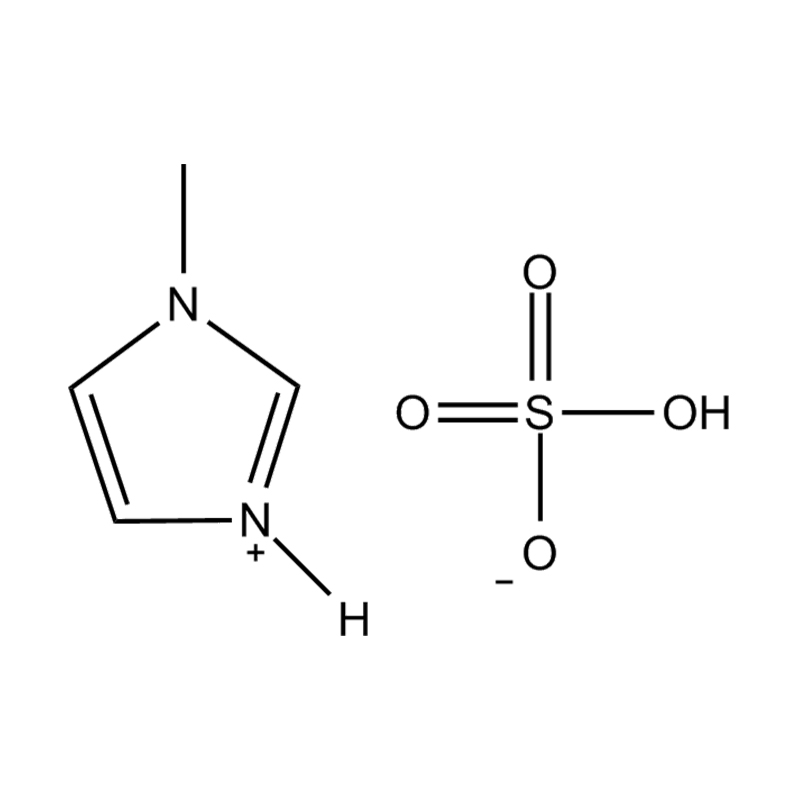
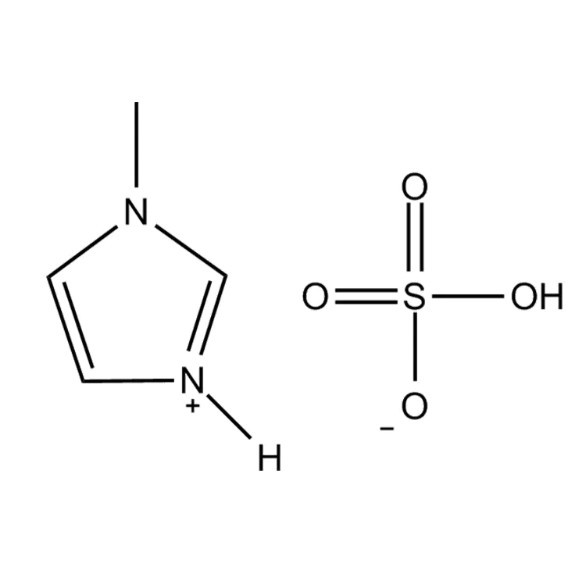
N-methylimidazolium hydrogen sulfate, commonly written as [Hmim][HSO₄], is a Brønsted acidic ionic liquid formed by the protonation of 1-methylimidazole with sulfuric acid. Unlike conventional ionic liquids that are typically formed through quaternization reactions, this compound retains an acidic proton on the imidazolium nitrogen, giving it a unique combination of ionic liquid characteristics and strong Brønsted acid functionality. It belongs to the broader family of protic ionic liquids (PILs), which are distinguished from aprotic ionic liquids by the presence of a transferable proton and the associated hydrogen-bonding network that this creates within the liquid structure.
The compound has attracted considerable research and industrial interest over the past two decades because it simultaneously functions as a solvent, a catalyst, and a reaction medium — roles that are typically distributed among multiple separate reagents in conventional chemistry. Its synthesis is straightforward and scalable, its toxicity profile is generally more favorable than many conventional acid catalysts, and its negligible vapor pressure minimizes worker exposure and atmospheric emissions. These features have made [Hmim][HSO₄] a subject of intensive investigation in green chemistry, biomass conversion, electrochemistry, and organic synthesis.
Chemical Identity and Structural Characteristics
The molecular structure of N-methylimidazolium hydrogen sulfate consists of a 1-methylimidazolium cation ([Hmim]⁺) paired with a hydrogen sulfate anion ([HSO₄]⁻). The cation is formed when the N-3 nitrogen of 1-methylimidazole accepts a proton from sulfuric acid, creating a positively charged aromatic ring with a methyl group at N-1 and a proton at N-3. The hydrogen sulfate anion retains one acidic hydrogen, making it capable of both hydrogen bond donation and acceptance, which significantly influences the bulk physical properties of the material.
This hydrogen bonding between the cation's N-H group and the anion's oxygen atoms creates an extended ionic network that raises the melting point relative to many imidazolium-based ionic liquids and contributes to the compound's relatively high viscosity at room temperature. The imidazolium ring itself is planar and aromatic, contributing to π–π stacking interactions that further structure the liquid phase at a molecular level. Understanding these structural features is essential for predicting how the compound will behave in different solvent systems and at different temperatures.
Key Physical and Chemical Properties
The physical and chemical properties of [Hmim][HSO₄] are directly relevant to its practical utility. The table below summarizes the most important documented values:
| Property | Reported Value / Description |
| Molecular Formula | C₄H₇N₂⁺ · HSO₄⁻ (C₄H₈N₂O₄S) |
| Molecular Weight | ~180.18 g/mol |
| Appearance | Colorless to pale yellow viscous liquid or solid |
| Melting Point | ~29–35°C (varies with purity and water content) |
| Decomposition Temperature | >200°C (thermally stable up to ~220°C) |
| Vapor Pressure | Negligible at ambient conditions |
| Viscosity (at 25°C) | Relatively high; decreases significantly with temperature |
| Solubility in Water | Fully miscible; highly hygroscopic |
| Acidity | Strong Brønsted acid; Hammett acidity function applicable |
| Electrical Conductivity | Moderate to high; suitable for electrochemical applications |
| Polarity | High polarity; dissolves polar and some non-polar substrates |
Thermal Stability and Liquid Range
The thermal stability of [Hmim][HSO₄] is one of its most operationally valuable properties. Thermogravimetric analysis (TGA) studies show that the compound begins to decompose at temperatures above approximately 200 to 220°C, giving it a wide liquid-phase operating window once it melts near room temperature. This broad temperature range is far wider than most conventional molecular solvents and allows reactions to be performed at elevated temperatures without the risk of solvent evaporation, reflux losses, or pressure buildup in closed systems. The low melting point — close to ambient temperature — means it can be handled as a liquid in most laboratory and industrial environments without preheating.
Brønsted Acidity and Proton-Transfer Behavior
The defining chemical property of [Hmim][HSO₄] is its strong Brønsted acidity, which arises from both the N-H proton on the imidazolium cation and the acidic proton of the hydrogen sulfate anion. This dual-source acidity gives the compound a higher effective proton availability compared to monoprotic acid-derived ionic liquids. The Hammett acidity function (H₀) values measured for this compound and related systems confirm acidity levels that are effective for proton-catalyzed reactions without reaching the superacid regime. This makes [Hmim][HSO₄] a controllable and selective acid catalyst, capable of promoting reactions that require significant proton activity without the uncontrolled reactivity and corrosivity associated with concentrated mineral acids.
Role as an Acid Catalyst in Organic Synthesis
The most extensively studied application of N-methylimidazolium hydrogen sulfate is as a Brønsted acid catalyst for organic reactions. In this role, it replaces conventional liquid acids such as sulfuric acid, hydrochloric acid, and p-toluenesulfonic acid while offering the additional benefits of recyclability, low volatility, and easier product separation. The ionic liquid phase and the organic product phase often spontaneously separate upon reaction completion, allowing the catalyst to be recovered by simple decantation and reused across multiple reaction cycles with minimal activity loss.
Key reaction types catalyzed effectively by [Hmim][HSO₄] include esterification and transesterification, Fischer indole synthesis, Beckmann rearrangement, Fries rearrangement, Friedel-Crafts acylation under mild conditions, and the synthesis of heterocyclic compounds including dihydropyrimidinones via the Biginelli reaction. In esterification reactions, the compound has shown catalytic activity comparable to concentrated sulfuric acid at equivalent acid loadings, while producing less byproduct formation and allowing straightforward workup. Its ability to function simultaneously as solvent and catalyst — in what is called a "solvent-catalyst" system — is particularly attractive because it eliminates the need for an additional inert solvent, reducing process complexity and waste generation.
Biomass Processing and Cellulose Dissolution
Among the most impactful emerging applications of [Hmim][HSO₄] is its use in the pretreatment and chemical conversion of lignocellulosic biomass. The conversion of agricultural waste, wood, and energy crops into fermentable sugars, platform chemicals, and biofuels requires breaking down the highly recalcitrant cellulose and hemicellulose matrix — a challenge that has historically demanded either expensive enzyme cocktails or harsh chemical treatments. Brønsted acidic ionic liquids based on the hydrogen sulfate anion have demonstrated an ability to disrupt hydrogen bonding networks in cellulose, facilitating its dissolution, hydrolysis, and subsequent conversion under relatively mild conditions.
Research groups have demonstrated that [Hmim][HSO₄] and related acidic ionic liquids can hydrolyze cellulose to glucose with yields exceeding 50 to 70 percent under optimized microwave or thermally assisted conditions, substantially outperforming dilute acid hydrolysis under equivalent conditions. The ionic liquid phase can also selectively dissolve hemicellulose while leaving lignin largely intact, enabling fractionation strategies that separately valorize each biomass component. The recyclability of the ionic liquid phase is a key economic advantage in biomass processing, as it offsets the higher initial cost of ionic liquid synthesis compared to mineral acid catalysts.
Biodiesel Synthesis and Esterification Catalysis
The production of biodiesel through acid-catalyzed esterification of free fatty acids (FFAs) is a specific area where [Hmim][HSO₄] has attracted strong commercial interest. Conventional base-catalyzed biodiesel processes are highly sensitive to the FFA content of the feedstock — when FFA levels exceed approximately 2 percent, soap formation and catalyst deactivation render the process uneconomical. Acid catalysts can handle high-FFA feedstocks, but traditional liquid acids create corrosion problems, require aqueous workup steps that generate wastewater, and cannot be easily recovered.
[Hmim][HSO₄] resolves these problems by providing strong Brønsted acidity in a non-corrosive, recoverable liquid catalyst format. Multiple studies have reported FFA conversion rates above 90 percent using this ionic liquid under moderate conditions (60–80°C, atmospheric pressure), with catalyst recycling demonstrated over five or more cycles without significant activity loss when properly dried between uses. The phase separation between the methanol-ester-glycerol product phase and the ionic liquid phase facilitates product recovery without aqueous washing steps, making the process considerably cleaner than conventional acid-catalyzed esterification routes.
Electrochemical Applications and Proton Conduction
The ionic conductivity and proton-transfer properties of [Hmim][HSO₄] make it a candidate electrolyte material for electrochemical devices, particularly proton exchange membrane fuel cells (PEMFCs) operating at intermediate temperatures (100–200°C). Conventional Nafion-based membranes in PEMFCs require continuous humidification and perform poorly above 80°C, creating engineering challenges for heat management and catalyst tolerance. Protic ionic liquids based on the imidazolium–hydrogen sulfate system exhibit proton conductivity through a Grotthuss-type mechanism involving proton hopping along the hydrogen-bonded ionic network, which remains active at temperatures well above 100°C without reliance on liquid water.
Research into composite membranes incorporating [Hmim][HSO₄] within polymer matrices has shown conductivity values in the range of 10⁻³ to 10⁻² S/cm at temperatures between 100 and 180°C — comparable to humidified Nafion in the same temperature range. This opens pathways to anhydrous or low-humidity PEMFC operation, which would simplify system design and improve tolerance to CO poisoning of platinum catalysts. Beyond fuel cells, the compound's conductivity and wide electrochemical window also make it attractive for use in supercapacitor electrolytes and electrodeposition media.
Handling, Safety, and Environmental Considerations
While ionic liquids are frequently described as "green" solvents due to their negligible volatility, the environmental and safety profile of [Hmim][HSO₄] must be evaluated in full context. The compound is strongly acidic and corrosive to skin and mucous membranes, requiring appropriate personal protective equipment including chemical-resistant gloves, eye protection, and adequate ventilation when handling. Its high hygroscopicity means water content must be carefully controlled in applications where anhydrous conditions are required, as absorbed moisture can significantly alter viscosity, melting point, and catalytic activity.
From an environmental standpoint, [Hmim][HSO₄] and structurally related imidazolium ionic liquids have been shown to exhibit aquatic toxicity toward certain microorganisms at higher concentrations, and biodegradation in conventional wastewater treatment systems is slow. Responsible use requires containment of process streams, avoidance of discharge into aquatic environments, and implementation of recovery and recycling protocols that maximize reuse and minimize disposal. The development of biodegradable ionic liquid analogs incorporating bio-based anions or cations is an active research direction aimed at addressing these concerns while preserving the functional advantages of the compound class.
Summary of Principal Uses
The versatility of N-methylimidazolium hydrogen sulfate across different application domains reflects its combination of strong Brønsted acidity, ionic liquid properties, thermal stability, and recyclability. The principal uses documented in the literature and in industrial practice include:
- Acid catalyst for esterification and biodiesel production from high-FFA feedstocks with straightforward phase separation and catalyst recovery.
- Solvent-catalyst for organic synthesis including Biginelli reactions, Fischer indole synthesis, and Friedel-Crafts transformations without additional solvent.
- Biomass pretreatment and cellulose hydrolysis for production of fermentable sugars and platform chemicals from lignocellulosic feedstocks.
- Electrolyte component in intermediate-temperature fuel cells and electrochemical devices requiring anhydrous proton conduction above 100°C.
- Reaction medium for heterocycle synthesis where the acidic ionic liquid environment promotes cyclization and condensation reactions with improved selectivity.
- Extractant and phase-transfer medium in separations chemistry, particularly for extracting polar compounds from aqueous systems or facilitating liquid–liquid biphasic reactions.
As research into ionic liquid chemistry continues to mature, [Hmim][HSO₄] remains one of the most frequently studied and practically deployed members of the Brønsted acidic ionic liquid family, owing to its accessible synthesis, well-characterized properties, and demonstrated performance across a uniquely broad range of chemical and electrochemical applications.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体