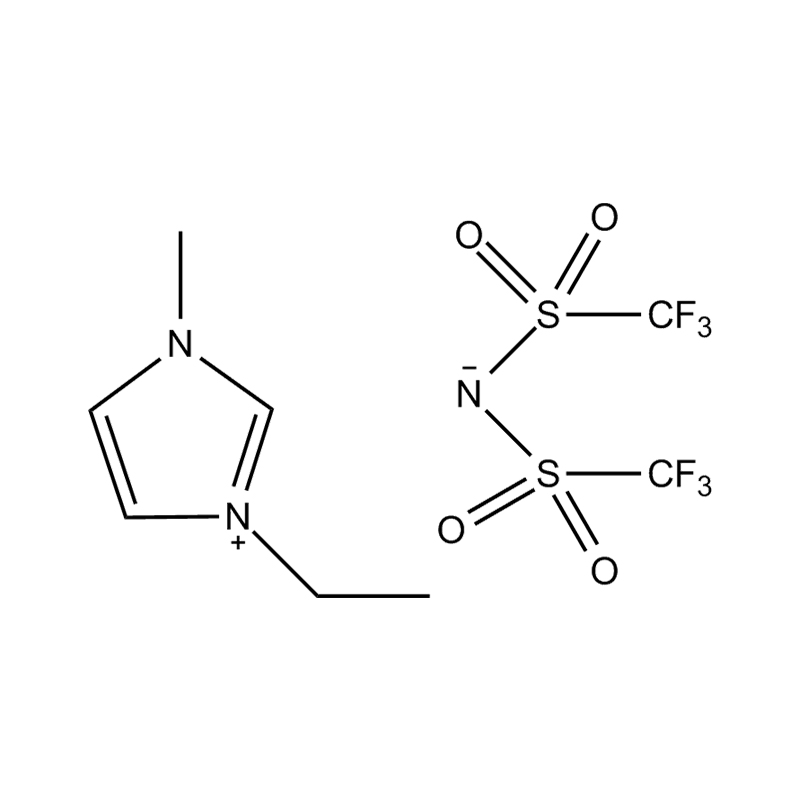
1-Ethyl-3-methylimidazolium bis(fluorosulfonyl)imide — commonly abbreviated as [EMIM][FSI] — is an ionic liquid that has attracted intensive scientific and industrial attention over the past two decades. Ionic liquids are salts that exist in liquid form at or near room temperature, and [EMIM][FSI] stands out within this broad family due to an exceptional combination of properties: very low viscosity, wide electrochemical stability window, high ionic conductivity, negligible vapor pressure, and good thermal stability. These characteristics make it one of the most versatile and practically useful ionic liquids available, with active applications spanning energy storage, electrochemical synthesis, lubrication science, and advanced materials research.
Content
- 1 Core Physical and Chemical Properties That Enable Its Uses
- 2 Electrolyte in Lithium-Ion and Next-Generation Batteries
- 3 Supercapacitor and Electrochemical Capacitor Electrolytes
- 4 Electrodeposition of Metals and Semiconductors
- 5 Use as a Solvent and Reaction Medium in Chemical Synthesis
- 6 Lubrication and Tribological Applications
- 7 Summary of Key Application Areas
- 8 Handling, Safety, and Practical Considerations
Core Physical and Chemical Properties That Enable Its Uses
Understanding why [EMIM][FSI] is so widely applied requires a clear picture of what makes it physically and chemically distinctive. The bis(fluorosulfonyl)imide anion — also written FSI⁻ — is a weakly coordinating, highly delocalized anion that interacts only loosely with the imidazolium cation. This weak ion pairing is the root cause of the compound's remarkably low viscosity compared to many other ionic liquids. At 25°C, [EMIM][FSI] has a dynamic viscosity of approximately 18–22 mPa·s, which is low enough to allow reasonable ion mobility without requiring elevated temperatures.
Its ionic conductivity at room temperature falls in the range of 14–18 mS/cm, among the highest recorded for any pure ionic liquid. This is a direct consequence of the low viscosity and the high charge density of the FSI⁻ anion. The electrochemical window — the voltage range across which the compound neither oxidizes nor reduces — spans approximately 4.5 to 5.5 V depending on electrode material and measurement conditions. This broad window is what makes [EMIM][FSI] so attractive as an electrolyte medium for high-voltage electrochemical applications. Its melting point is well below 0°C (reported values range from −18°C to −22°C), meaning it remains liquid across most operational temperature ranges relevant to real-world devices.
Electrolyte in Lithium-Ion and Next-Generation Batteries
The most commercially significant application of [EMIM][FSI] is as an electrolyte component in rechargeable battery systems. Conventional lithium-ion batteries use organic carbonate electrolytes — ethylene carbonate, dimethyl carbonate, and related compounds — which are flammable and prone to decomposition at elevated temperatures or after cell abuse. Ionic liquids offer a non-flammable, thermally stable alternative, and [EMIM][FSI] is among the most suitable candidates because its low viscosity allows lithium ions to migrate through the electrolyte at rates fast enough for practical charge and discharge cycling.
In lithium battery research, [EMIM][FSI] is typically used as a host solvent into which a lithium salt — most commonly lithium bis(fluorosulfonyl)imide (LiFSI) — is dissolved at concentrations between 0.5 M and 3.2 M. At high lithium salt concentrations, the electrolyte forms a "locally concentrated" ionic liquid electrolyte with improved compatibility with graphite anodes, which would otherwise be exfoliated by the imidazolium cation. Studies have demonstrated stable cycling of graphite/LiFePO₄ and graphite/NMC full cells using [EMIM][FSI]-based electrolytes at temperatures from −20°C to 60°C, outperforming carbonate electrolytes at both extremes of this range.
Sodium-Ion and Potassium-Ion Battery Applications
Beyond lithium, [EMIM][FSI] is being actively investigated as an electrolyte medium for sodium-ion and potassium-ion batteries — two post-lithium chemistries being developed as lower-cost alternatives for stationary energy storage. Sodium and potassium salts of the FSI⁻ anion dissolve readily in [EMIM][FSI], and the resulting electrolytes support reversible plating and stripping of these metals at conditions that are difficult to achieve in standard carbonate or ether-based solvents. The non-flammable nature of the ionic liquid electrolyte is particularly attractive for large-format stationary storage where fire safety is a primary design constraint.
Supercapacitor and Electrochemical Capacitor Electrolytes
Electrochemical double-layer capacitors (EDLCs), commonly called supercapacitors or ultracapacitors, store energy by adsorbing ions at the surface of high-surface-area carbon electrodes. The maximum energy density achievable in an EDLC scales with the square of the operating voltage, which means expanding the voltage window directly multiplies the energy stored per unit mass. Aqueous electrolytes limit EDLC operation to roughly 1 V, while organic electrolytes extend this to about 2.7 V. [EMIM][FSI], with its electrochemical window exceeding 4 V in carbon electrode cells, enables EDLC devices to operate at 3.5 V or higher, nearly doubling the achievable energy density compared to acetonitrile-based organic electrolytes.
The low viscosity of [EMIM][FSI] is critical in this context because it allows ions to penetrate the narrow pores of activated carbon and carbide-derived carbon electrode materials efficiently, even at sub-ambient temperatures. Research groups have demonstrated [EMIM][FSI]-based EDLC cells with specific energy values exceeding 40 Wh/kg at the device level — a benchmark that approaches the lower performance range of lead-acid batteries while maintaining the power density and cycle life advantages characteristic of capacitor-type storage.
Electrodeposition of Metals and Semiconductors
Electrodeposition — the process of reducing metal ions from solution onto an electrode surface to form a thin film or coating — is severely constrained in aqueous electrolytes because water electrolyzes below 1.23 V. Many metals of industrial interest, including aluminum, titanium, silicon, germanium, and refractory metals such as tantalum and niobium, cannot be electrodeposited from water at all because their reduction potentials lie below the hydrogen evolution limit. [EMIM][FSI] dissolves appropriate precursor salts for several of these elements and provides the electrochemical window needed to reduce them without competing electrolyte decomposition reactions.
Aluminum electrodeposition from [EMIM][FSI]-based electrolytes containing aluminum chloride (AlCl₃) has been demonstrated at room temperature with good current efficiency and controllable film morphology. The deposited aluminum coatings show promise for corrosion protection applications where conventional aqueous chromate or nickel plating is being phased out for environmental reasons. Silicon and germanium thin films deposited from [EMIM][FSI]-based electrolytes have been explored as anode materials for battery applications, where the electrodeposition route offers an alternative to high-temperature vacuum deposition methods.
Semiconductor and Nanostructure Synthesis
The unique solvation environment of [EMIM][FSI] also enables the synthesis of semiconductor nanostructures — quantum dots, nanowires, and thin films — with controlled morphology and composition. The ionic liquid acts simultaneously as solvent, structure-directing agent, and electrochemical medium, guiding the nucleation and growth of deposited materials through its organized interfacial structure at electrode surfaces. Compound semiconductors such as CdTe and Cu₂ZnSnS₄ (CZTS), relevant to solar cell manufacturing, have been deposited from [EMIM][FSI]-based electrolytes with compositional control not easily achieved in aqueous systems.
Use as a Solvent and Reaction Medium in Chemical Synthesis
Ionic liquids have been promoted as "green" alternatives to volatile organic solvents in chemical synthesis because their negligible vapor pressure eliminates solvent emission during reactions. [EMIM][FSI] participates in this application space, particularly for reactions that benefit from its specific solvation properties or where its electrochemical stability allows it to be used as a combined solvent and electrolyte for electrosynthesis.
Organic electrosynthesis — using electricity rather than chemical oxidants or reductants to drive organic transformations — is an area of growing industrial interest for producing pharmaceutical intermediates and fine chemicals. [EMIM][FSI] functions as both solvent and supporting electrolyte in such reactions, eliminating the need to dissolve a separate salt in an organic solvent and simplifying downstream product isolation. Its low viscosity relative to other ionic liquids improves mass transport within the electrochemical reactor, increasing current efficiency and reducing reaction times.
In CO₂ electrochemical reduction — a reaction of significant interest for converting captured carbon dioxide into useful fuels or chemicals — [EMIM][FSI] has been identified as a highly effective medium. The imidazolium cation participates actively in stabilizing the CO₂ radical anion intermediate, lowering the overpotential required for CO₂ reduction and improving the selectivity toward carbon monoxide or formate products compared to aqueous electrolytes.
Lubrication and Tribological Applications
The thermal stability, non-volatility, and tunable surface affinity of [EMIM][FSI] make it a viable lubricant additive and neat lubricant for demanding tribological applications. Unlike petroleum-based lubricants, it does not evaporate under vacuum conditions, making it suitable for use in space mechanisms, vacuum chambers, and precision instrument bearings where outgassing must be minimized. Studies of [EMIM][FSI] as a lubricant on steel-on-steel sliding contacts have shown significant reductions in friction coefficient and wear volume compared to unlubricated surfaces and to reference mineral oil lubricants.
The FSI⁻ anion contributes to tribological performance by forming a protective tribofilm on metal surfaces under shear conditions. The fluorine content of the anion plays a role analogous to that of PTFE (polytetrafluoroethylene) particles in conventional lubricant formulations, providing a low-energy surface chemistry that reduces adhesive wear. For aluminum alloys and soft metals that are difficult to protect with sulfur-phosphorus additive chemistry (which can corrode non-ferrous surfaces), [EMIM][FSI] offers a chemically compatible alternative.
Summary of Key Application Areas
The table below consolidates the primary uses of [EMIM][FSI] alongside the specific property that makes it suitable for each application domain.
| Application | Key Property Utilized | Performance Highlight |
|---|---|---|
| Li/Na/K-ion battery electrolyte | High ionic conductivity, non-flammability | Stable cycling from −20°C to 60°C |
| Supercapacitor electrolyte | Wide electrochemical window, low viscosity | Operating voltage >3.5 V; energy density >40 Wh/kg |
| Metal and semiconductor electrodeposition | Wide electrochemical window, negligible water | Enables Al, Si, Ge deposition at room temperature |
| Electrosynthesis and CO₂ reduction | Cation-mediated intermediate stabilization | Reduced overpotential; improved CO selectivity |
| Lubrication (vacuum/precision systems) | Zero vapor pressure, thermal stability | Viable in vacuum; protective FSI-derived tribofilm |
Handling, Safety, and Practical Considerations
While [EMIM][FSI] is far less hazardous than the volatile organic solvents it often replaces, it is not without handling requirements. The compound is hygroscopic — it absorbs water from ambient air — and dissolved water affects its electrochemical window, viscosity, and conductivity. For electrochemical applications requiring performance at the limits of the stability window, [EMIM][FSI] should be dried under vacuum at 60–80°C with stirring until water content is below 20 ppm as measured by Karl Fischer titration.
- Store in sealed containers under inert atmosphere (argon or nitrogen) to minimize moisture absorption and prevent any reaction with atmospheric CO₂ which can alter ionic liquid composition over extended periods.
- Avoid prolonged skin contact — while [EMIM][FSI] has low acute toxicity, ionic liquids as a class show biological activity at cellular level, and cumulative exposure data is still being gathered by occupational health researchers.
- Handle glassware and equipment used with [EMIM][FSI] carefully — its low surface tension means it wets surfaces aggressively and can be difficult to remove completely from porous or roughened surfaces without thorough solvent washing.
- Disposal should follow local regulations for fluorine-containing chemicals — the FSI⁻ anion contains fluorosulfonyl groups that produce fluoride-containing byproducts upon incineration and should not be disposed of in standard aqueous waste streams without appropriate treatment.
As research into ionic liquids continues to mature and scale-up pathways for [EMIM][FSI] production become more cost-effective, the gap between laboratory performance and commercial deployment is closing steadily. Its combination of electrochemical breadth, low viscosity, and thermal robustness positions it as one of the most technically justified ionic liquids for transition from academic research into industrial practice across multiple sectors.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体