Content
What Is 1-Octyl-3-methylimidazolium Hexafluorophosphate?
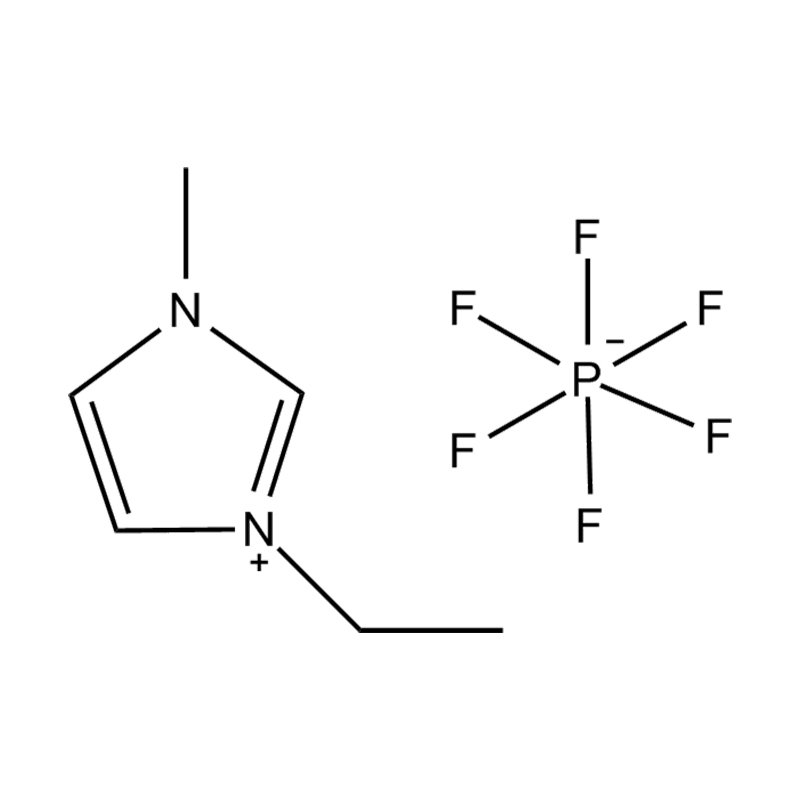
1-Octyl-3-methylimidazolium hexafluorophosphate, commonly abbreviated as [C8mim][PF6] or [OMIM][PF6], is a room-temperature ionic liquid (RTIL) that has attracted significant attention across chemistry, materials science, and industrial engineering. It belongs to the imidazolium-based family of ionic liquids and is characterized by its negligible vapor pressure, wide liquid temperature range, excellent thermal stability, and tunable physicochemical properties. With a molecular formula of C14H27F6N2P and a molecular weight of approximately 396.35 g/mol, this compound exists as a viscous, colorless to slightly yellow liquid at ambient conditions, making it particularly versatile as a solvent, electrolyte, or functional material in numerous advanced applications.
The compound is synthesized by alkylating 1-methylimidazole with 1-chlorooctane or 1-bromooctane to form the intermediate [C8mim]Cl or [C8mim]Br, followed by anion exchange with hexafluorophosphate salts such as KPF6 or HPF6. The resulting ionic liquid is hydrophobic due to the bulky PF6⁻ anion, which limits water miscibility and makes it highly suitable for liquid–liquid extraction and biphasic reaction systems.
Core Working Principles of [C8mim][PF6]
Understanding why [C8mim][PF6] performs so effectively in various fields requires a closer look at its fundamental physicochemical mechanisms. Its behavior arises from the interplay of ionic interactions, hydrogen bonding, van der Waals forces, and the amphiphilic structure imparted by the octyl chain.
Ion Pair Structure and Electrostatic Interactions
As a purely ionic compound in the liquid state, [C8mim][PF6] consists of discrete cation–anion pairs held together by electrostatic forces rather than covalent bonds. The imidazolium cation carries a delocalized positive charge across its aromatic ring, while the PF6⁻ anion provides a diffuse negative charge due to the symmetrical arrangement of six fluorine atoms around a central phosphorus. This charge distribution results in moderate ion-pairing strength, which enables reasonable ionic conductivity while maintaining liquid fluidity at room temperature. The weak coordination of PF6⁻ with the cation also contributes to the ionic liquid's electrochemical stability window, typically exceeding 4.5 V, making it superior to many conventional electrolytes.
Hydrophobicity from the Octyl Chain and PF6⁻ Anion
The eight-carbon alkyl chain on the imidazolium cation provides substantial nonpolar character, and when combined with the hydrophobic PF6⁻ anion, the resulting liquid is largely immiscible with water under ambient conditions. This hydrophobicity is a critical working mechanism in extraction processes, as it allows [C8mim][PF6] to form a distinct organic-like phase that selectively partitions target analytes away from aqueous matrices. The longer chain length compared to shorter imidazolium counterparts (e.g., [C4mim][PF6]) enhances hydrophobicity and increases viscosity, which affects mass transfer kinetics but improves selectivity in separation applications.
Thermal and Chemical Stability
The thermal decomposition temperature of [C8mim][PF6] is typically above 300 °C, and it remains liquid from well below 0 °C up to its decomposition point. This exceptionally wide liquid range allows it to function in high-temperature reaction environments where conventional organic solvents would evaporate or decompose. Its resistance to oxidation and reduction under a broad electrochemical window further establishes the compound as a stable medium for both chemical synthesis and electrochemical measurements.
Key Application Scenarios
The unique combination of properties described above gives rise to an impressive array of applications spanning analytical chemistry, energy storage, catalysis, tribology, and environmental remediation.
Electrochemical Sensing and Biosensors
One of the most actively researched applications of [C8mim][PF6] is in electrochemical sensing. When incorporated into modified electrode surfaces or used as a binder in carbon paste electrodes, it enhances electron transfer kinetics by acting as a highly conductive, non-volatile medium that keeps the electrode surface active. Sensors based on [C8mim][PF6]-modified electrodes have demonstrated improved sensitivity for the detection of heavy metals (such as Pb²⁺, Cd²⁺, and Hg²⁺), pharmaceutical compounds, pesticides, and biological analytes like dopamine and ascorbic acid. The ionic liquid's viscous nature helps immobilize enzymes or nanoparticles on electrode surfaces without degrading their activity, making it a useful component in third-generation biosensors.
Liquid–Liquid Extraction of Pollutants and Metals
The hydrophobic biphasic behavior of [C8mim][PF6] makes it a powerful solvent for dispersive liquid–liquid microextraction (DLLME) and solvent extraction systems. In environmental analysis, it has been applied for the extraction and preconcentration of polycyclic aromatic hydrocarbons (PAHs), organochlorine pesticides, endocrine-disrupting compounds, and trace heavy metals from water samples prior to chromatographic analysis. The extraction mechanism relies on favorable partition coefficients: hydrophobic or ion-pairable target molecules preferentially migrate into the [C8mim][PF6] phase, achieving enrichment factors often exceeding 100-fold. In metal recovery, ionic liquid phases can be functionalized or combined with chelating agents to selectively extract valuable metals like gold, platinum, or uranium from leachates.
Electrolyte in Batteries and Supercapacitors
[C8mim][PF6] is employed as an electrolyte or co-solvent in lithium-ion batteries, electric double-layer capacitors (EDLCs), and dye-sensitized solar cells (DSSCs). Its wide electrochemical window prevents decomposition at high operating voltages, and its non-flammability addresses critical safety concerns associated with conventional organic electrolytes. In supercapacitors, it enables efficient charge storage through the reversible adsorption of ions at high-surface-area carbon electrodes. Researchers have also explored its role in quasi-solid-state electrolytes, where [C8mim][PF6] is embedded into polymer matrices to improve mechanical flexibility while retaining ionic conductivity.
Green Solvent for Synthesis and Catalysis
As a designer solvent, [C8mim][PF6] has been used to replace volatile organic compounds (VOCs) in organic synthesis, aligning with green chemistry principles. It has been demonstrated as an effective medium for Diels–Alder reactions, Heck couplings, Friedel–Crafts alkylations, and enzymatic transformations. The ionic environment stabilizes polar transition states, often accelerating reaction rates and improving selectivity compared to molecular solvents. Because the ionic liquid phase can be recycled multiple times without significant performance loss, it reduces solvent waste — a major advantage in industrial-scale synthesis.
Lubrication and Tribological Applications
The high thermal stability, low volatility, and film-forming capability of [C8mim][PF6] make it a strong candidate for advanced lubricants and anti-wear coatings. Studies have shown that when used as a neat lubricant or additive in base oils, it significantly reduces friction coefficients and wear rates on steel, aluminum, and silicon surfaces. The electrostatic adsorption of the imidazolium cation onto negatively charged metal surfaces forms a protective ionic layer that withstands high contact pressures. This application is particularly relevant in aerospace and vacuum environments, where conventional lubricants fail due to evaporation.
Comparative Properties Relevant to Application Selection
Selecting [C8mim][PF6] over other ionic liquids or conventional solvents involves weighing several key parameters. The following table summarizes important properties that guide application-specific decision-making:
| Property | Value / Characteristic | Application Relevance |
| Electrochemical window | ~4.5 – 5.0 V | Energy storage, sensors |
| Viscosity (25 °C) | ~500–700 mPa·s | Lubrication, electrode binder |
| Thermal stability | >300 °C | High-temp catalysis, lubrication |
| Water miscibility | Hydrophobic (immiscible) | Liquid–liquid extraction |
| Vapor pressure | Negligible | Green solvent, vacuum use |
Limitations and Considerations in Practical Use
Despite its many advantages, [C8mim][PF6] presents several practical limitations that must be considered when designing processes around it. Its high viscosity at room temperature can hinder mass transfer in extraction and catalytic systems, often necessitating mild heating or the use of co-solvents to improve fluidity. The PF6⁻ anion, while contributing to hydrophobicity and electrochemical stability, is susceptible to slow hydrolysis under acidic or basic aqueous conditions, releasing fluoride ions and potentially causing corrosion of metal equipment.
From an environmental perspective, imidazolium-based ionic liquids including [C8mim][PF6] have been found to exhibit moderate ecotoxicity, particula


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体