Content
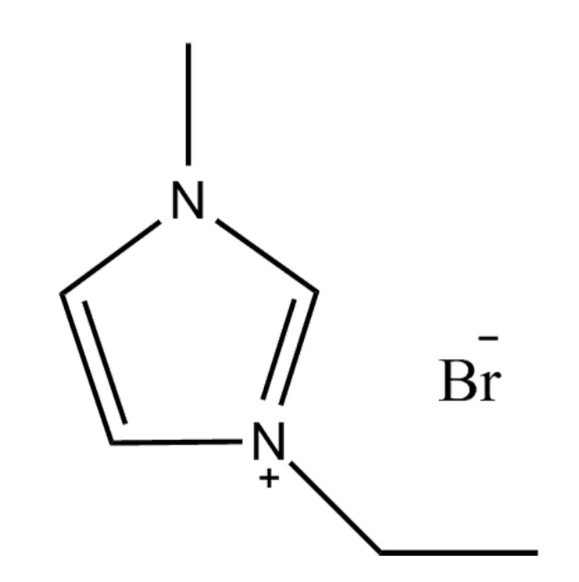
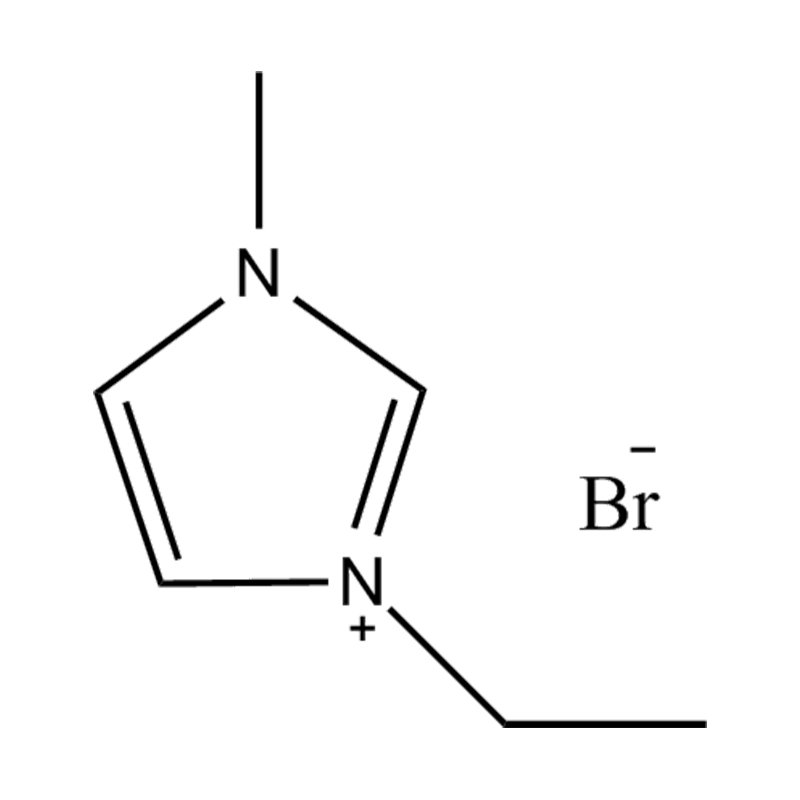
Introduction to 1-Ethyl-3-methylimidazolium Bromide
1-Ethyl-3-methylimidazolium bromide is a type of ionic liquid known for its unique chemical stability, low volatility, and excellent solubility characteristics. These properties make it a valuable component in chemical synthesis, catalysis, and electrochemical applications. As a room-temperature ionic liquid, it provides an environmentally friendlier alternative to traditional organic solvents.
Understanding its solubility and practical uses can help researchers and industrial chemists design more efficient processes and select the right ionic liquids for specific reactions or applications.
Chemical Properties and Solubility Characteristics
Molecular Structure
The molecule consists of an imidazolium cation with ethyl and methyl substituents and a bromide anion. This structure facilitates strong ionic interactions and hydrogen bonding, contributing to its high solubility in both polar and certain nonpolar solvents. Its polarity also makes it miscible with water and many organic solvents such as methanol, ethanol, and acetonitrile.
Solubility in Various Solvents
1-Ethyl-3-methylimidazolium bromide exhibits:
- High solubility in polar solvents due to strong electrostatic interactions.
- Moderate solubility in less polar organic solvents, enabling it to act as a phase-transfer medium.
- Water miscibility, making it suitable for aqueous-phase reactions and green chemistry applications.
Thermal and Chemical Stability
This ionic liquid remains stable across a wide temperature range, typically up to 250°C, and resists hydrolysis and decomposition under neutral or slightly acidic conditions. Its chemical inertness makes it an ideal medium for catalytic reactions where traditional solvents may degrade or evaporate.
Applications in Industrial Chemistry
Catalysis
1-Ethyl-3-methylimidazolium bromide serves as an effective solvent and co-catalyst in many organic reactions. Its ability to dissolve both organic and inorganic compounds enhances reaction rates and selectivity. For example, it is often used in:
- Friedel–Crafts alkylation and acylation reactions.
- Carbon–carbon bond forming reactions like Suzuki and Heck couplings.
- Oxidation reactions with metal catalysts, where solubility of both the substrate and catalyst is crucial.
Electrochemistry
Due to its ionic nature and wide electrochemical window, it is widely applied in electrochemical devices such as:
- Electroplating and electrodeposition processes.
- Electrochemical sensors and batteries, where stability and conductivity are essential.
- Green energy systems, including supercapacitors.
Extraction and Separation Processes
Its high solubility and selective interactions with metal ions allow it to be used in:
- Metal recovery and purification from aqueous solutions.
- Removal of organic contaminants from wastewater streams.
- Phase-transfer catalysis where hydrophobic and hydrophilic components need to be separated efficiently.
Biomedical and Material Science Uses
Pharmaceutical Applications
1-Ethyl-3-methylimidazolium bromide is increasingly investigated for its ability to improve solubility of poorly soluble drugs, providing:
- Enhanced drug delivery in aqueous environments.
- Stabilization of sensitive biomolecules against hydrolysis or denaturation.
- Support in enzymatic reactions due to its mild and non-volatile nature.
Material Synthesis
In material science, it is utilized for:
- Preparation of polymeric ionic liquids and composite materials.
- Controlled crystallization of salts and metal-organic frameworks.
- Stabilizing nanoparticles and catalysts for improved surface activity.
Safety and Handling Considerations
Toxicity Profile
While generally considered less volatile and safer than many organic solvents, standard safety precautions must be observed. It is recommended to:
- Use gloves and eye protection when handling concentrated forms.
- Avoid inhalation and prolonged skin contact.
- Store in tightly sealed containers to prevent moisture absorption and contamination.
Environmental Impact
Due to its negligible vapor pressure, it reduces air emissions, making it a green alternative to volatile organic solvents. However, it should be properly recycled or disposed of according to local regulations to minimize environmental accumulation.
Conclusion
1-Ethyl-3-methylimidazolium bromide combines unique solubility, chemical stability, and versatility, making it a highly useful ionic liquid for industrial chemistry, electrochemistry, material science, and pharmaceutical applications. Its properties allow for safer, more efficient, and environmentally friendly processes compared to traditional organic solvents.
Careful consideration of solubility characteristics and application requirements ensures optimal performance. With proper handling, this ionic liquid can significantly improve reaction efficiency, product quality, and overall process sustainability.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体