Content
- 1 What Is 1-Ethyl-3-Methylimidazolium Trifluoromethanesulfonate?
- 2 Key Physical and Chemical Properties
- 3 Synthesis and Purification Methods
- 4 Electrochemical Applications: Electrolytes and Energy Storage
- 5 Catalysis and Organic Synthesis Applications
- 6 Applications in Materials Science and Nanotechnology
- 7 Safety, Handling, and Environmental Considerations
- 8 Selecting [EMIM][OTf] for Your Application: Key Decision Criteria
What Is 1-Ethyl-3-Methylimidazolium Trifluoromethanesulfonate?
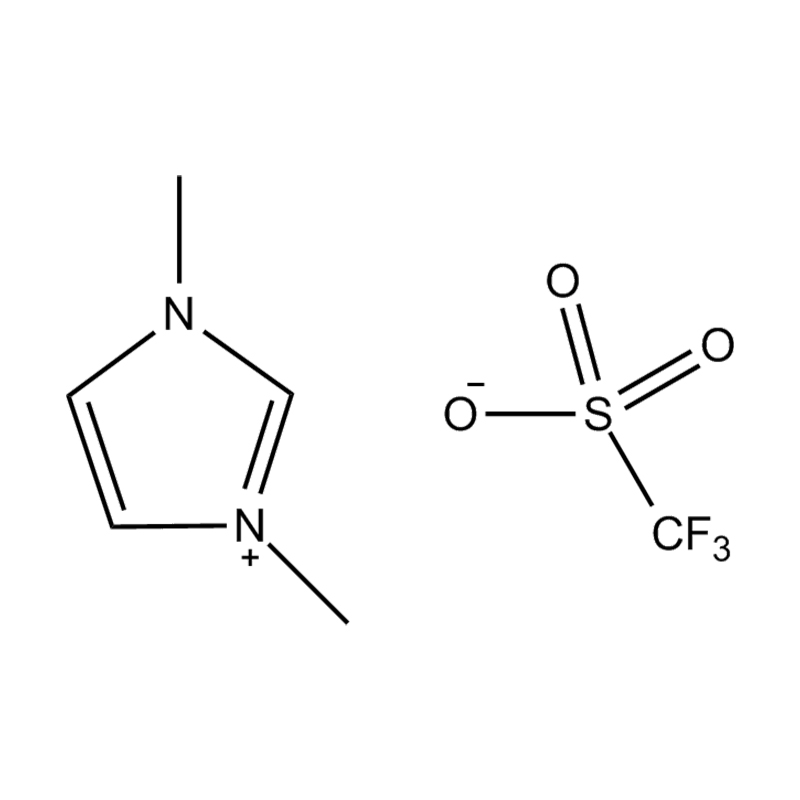
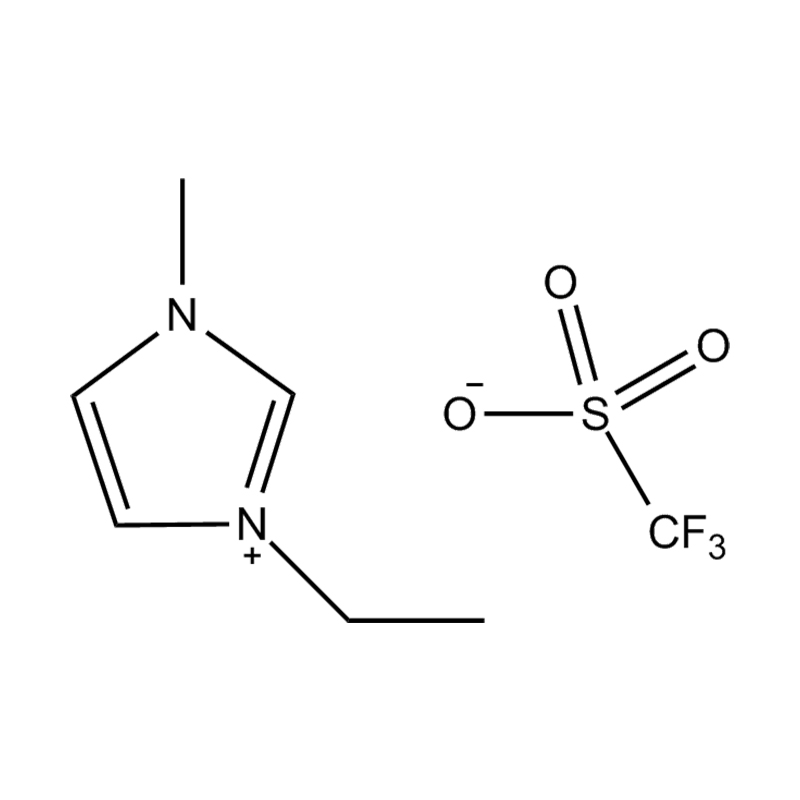
1-Ethyl-3-methylimidazolium trifluoromethanesulfonate, commonly abbreviated as [EMIM][OTf] or EMIMOTf, is a room-temperature ionic liquid (RTIL) belonging to the imidazolium family — one of the most extensively studied and commercially significant classes of ionic liquids in modern chemistry. Its IUPAC name reflects its two-ion architecture: a 1-ethyl-3-methylimidazolium cation paired with a trifluoromethanesulfonate (triflate) anion. The compound carries the CAS registry number 145022-44-2 and has a molecular formula of C₇H₁₁F₃N₂O₃S, with a molecular weight of approximately 260.23 g/mol. Unlike conventional organic solvents, [EMIM][OTf] exists as a liquid at or near room temperature despite being composed entirely of ions, a property that distinguishes ionic liquids from both traditional molten salts and molecular solvents and underpins their remarkable versatility as functional materials.
The triflate anion (CF₃SO₃⁻) is a weakly coordinating, highly stable anion that imparts a distinctive set of physicochemical properties to the ionic liquid — including low viscosity relative to many other imidazolium salts, broad electrochemical stability, excellent thermal resistance, and high ionic conductivity. These characteristics have driven substantial academic and industrial interest in [EMIM][OTf] as a solvent, electrolyte, catalyst medium, and functional material across disciplines ranging from electrochemistry and materials science to pharmaceutical synthesis and green chemistry.
Key Physical and Chemical Properties
Understanding the specific physicochemical properties of [EMIM][OTf] is essential for evaluating its suitability for any given application. The compound's properties are well-characterized in the scientific literature and represent a favorable combination of stability, conductivity, and processability that distinguishes it from many competing ionic liquids.
| Property | Value / Description |
| Molecular Formula | C₇H₁₁F₃N₂O₃S |
| Molecular Weight | 260.23 g/mol |
| Melting Point | ~ -9°C (liquid at room temperature) |
| Thermal Decomposition Temperature | > 400°C |
| Viscosity (25°C) | ~ 43–45 mPa·s |
| Ionic Conductivity (25°C) | ~ 8–9 mS/cm |
| Electrochemical Window | ~ 4.1–4.3 V |
| Vapor Pressure | Negligible at ambient conditions |
| Appearance | Colorless to pale yellow liquid |
| Solubility in Water | Miscible |
The negligible vapor pressure of [EMIM][OTf] is one of its most practically significant properties. Conventional organic solvents such as acetonitrile, dichloromethane, and diethyl ether evaporate readily at ambient conditions, creating volatile organic compound (VOC) emissions that pose health risks, fire hazards, and environmental concerns. Because [EMIM][OTf] exerts essentially no vapor pressure under normal operating conditions, it does not evaporate, eliminating solvent loss during reactions, simplifying product isolation through evaporation, and dramatically reducing airborne exposure risks in laboratory and industrial settings.
Synthesis and Purification Methods
The synthesis of [EMIM][OTf] is straightforward relative to many specialty chemicals and can be accomplished through well-established metathesis and direct alkylation routes. The most direct synthetic route involves the quaternization of 1-methylimidazole with ethyl trifluoromethanesulfonate (ethyl triflate) in a single-step reaction. When 1-methylimidazole is combined with ethyl triflate — a highly reactive alkylating agent — the nitrogen atom at the 3-position of the imidazole ring undergoes N-alkylation, directly yielding the [EMIM][OTf] ionic liquid without requiring an anion exchange step.
An alternative two-step route first prepares 1-ethyl-3-methylimidazolium halide (typically the chloride or bromide salt) by reacting 1-methylimidazole with an ethyl halide, then performs an anion exchange reaction by treating the halide salt with a silver triflate, lithium triflate, or triflic acid solution to replace the halide anion with the triflate anion. While this route avoids the use of the hazardous ethyl triflate reagent, it introduces the challenge of removing residual halide impurities, which must be reduced to sub-ppm levels for electrochemical applications where halide contamination causes significant degradation of performance.
Purification of [EMIM][OTf] typically involves the following steps to ensure research or application-grade purity:
- Washing with activated carbon in acetonitrile solution to remove colored organic impurities and trace starting materials
- Filtration through neutral alumina or silica gel columns to remove polar impurities and residual metal ions
- Rotary evaporation under reduced pressure to remove volatile solvents used in the purification steps
- Drying under high vacuum at elevated temperature (typically 60–80°C for 24–48 hours) to reduce water content to below 20 ppm for moisture-sensitive applications
- Halide content verification by ion chromatography or silver nitrate titration to confirm removal below the application-specific threshold
Water content management is particularly critical for [EMIM][OTf] intended for electrochemical use, as absorbed moisture significantly reduces the electrochemical window, increases conductivity through proton transport mechanisms that distort performance data, and can hydrolyze sensitive electrode materials or dissolved species. Dried [EMIM][OTf] should be stored under inert atmosphere (argon or nitrogen) in sealed containers to prevent reabsorption of atmospheric moisture.
Electrochemical Applications: Electrolytes and Energy Storage
The electrochemical properties of [EMIM][OTf] make it one of the most actively researched ionic liquid electrolytes for advanced energy storage and conversion devices. Its combination of wide electrochemical stability window (~4.1–4.3 V), high ionic conductivity (~8–9 mS/cm at room temperature), negligible volatility, and thermal stability up to over 400°C addresses several fundamental limitations of conventional electrolytes based on organic carbonate solvents, which are flammable, volatile, and restricted to electrochemical windows of approximately 4–5 V in practice.
Supercapacitors and Electric Double-Layer Capacitors
In electric double-layer capacitors (EDLCs), the energy storage mechanism relies on electrostatic ion adsorption at the electrode-electrolyte interface rather than faradaic chemical reactions. [EMIM][OTf] has been extensively evaluated as an EDLC electrolyte due to its favorable ion size, which allows effective penetration into the microporous structure of activated carbon electrodes, and its wide electrochemical window, which permits operation at higher cell voltages than aqueous electrolytes allow. Higher operating voltage directly increases energy density (which scales with the square of voltage), making ionic liquid electrolytes like [EMIM][OTf] central to next-generation high-energy-density supercapacitor development. Research groups have demonstrated [EMIM][OTf]-based EDLCs operating stably at cell voltages of 3.5 V or above, compared to the 1.0–1.2 V limit of aqueous systems.
Lithium-Ion and Sodium-Ion Battery Electrolytes
Mixtures of [EMIM][OTf] with lithium triflate or sodium triflate have been investigated as safer alternatives to conventional flammable carbonate electrolytes in lithium-ion and sodium-ion batteries. The non-flammability and thermal stability of [EMIM][OTf]-based electrolytes directly addresses the thermal runaway safety concern that has driven significant attention to battery safety in electric vehicle applications. Challenges remain in optimizing the solid electrolyte interphase (SEI) formed on lithium metal and graphite anodes in ionic liquid electrolytes, and in reducing viscosity at low temperatures where [EMIM][OTf] becomes significantly more viscous and ionic conductivity drops — an area of active materials engineering research.
Catalysis and Organic Synthesis Applications
[EMIM][OTf] has found productive application as a reaction medium and co-catalyst in a range of organic synthesis and catalytic transformation contexts, where its properties as a polar, non-coordinating solvent with negligible vapor pressure offer practical advantages over conventional organic solvents.
Acid-Catalyzed Reactions
The triflate anion is derived from triflic acid — one of the strongest known Brønsted acids — and [EMIM][OTf] can exhibit mild Lewis acid character under certain conditions, particularly in combination with metal triflate catalysts. It has been used as a co-solvent and activating medium in Friedel-Crafts alkylations, Diels-Alder cycloadditions, and glycosylation reactions, where its polarity stabilizes charged transition states and ion pairs, accelerating reaction rates and in some cases improving selectivity compared to conventional molecular solvents.
Transition Metal-Catalyzed Reactions
Palladium, ruthenium, and rhodium catalysts dissolved or immobilized in [EMIM][OTf] have been applied to cross-coupling reactions, hydrogenations, and carbonylation chemistry. The ionic liquid phase immobilizes the catalyst, facilitating product separation by extraction with non-polar solvents while retaining the metal catalyst in the ionic liquid phase for reuse over multiple reaction cycles — a biphasic catalysis strategy that addresses the challenge of expensive noble metal catalyst recovery and recycling in fine chemical synthesis.
Enzymatic and Biocatalytic Processes
A growing body of research has demonstrated that certain enzymes retain significant catalytic activity when dissolved or suspended in [EMIM][OTf] or [EMIM][OTf]-water mixtures. Lipases, proteases, and oxidoreductases have all been studied in this context, with [EMIM][OTf]'s relatively low viscosity and water miscibility proving advantageous for maintaining enzyme accessibility to substrates. The ability to dissolve both hydrophilic and hydrophobic substrates in a single ionic liquid phase — avoiding the phase partitioning challenges of biphasic aqueous-organic systems — represents a meaningful practical advantage in biocatalytic synthesis of pharmaceutical intermediates and fine chemicals.
Applications in Materials Science and Nanotechnology
[EMIM][OTf] has been adopted as a functional medium in a range of materials synthesis and nanotechnology applications, where its unique combination of properties enables processes and material structures difficult or impossible to achieve with conventional solvents.
- Electrodeposition of metals and semiconductors: The wide electrochemical window of [EMIM][OTf] allows electrodeposition of metals such as aluminum, titanium, and silicon that cannot be deposited from aqueous electrolytes due to competing water reduction reactions. This enables ionic liquid electrodeposition as a route to functional metal coatings, alloys, and semiconductor thin films for microelectronics and photovoltaic applications.
- Nanoparticle synthesis: [EMIM][OTf] acts as both a solvent and a stabilizing medium for metal nanoparticle synthesis, where its high viscosity relative to water and strong ion-pair interactions with nanoparticle surfaces help control nucleation and growth kinetics, producing nanoparticles with narrower size distributions than those obtained in conventional solvents.
- Polymer electrolytes and gel electrolytes: [EMIM][OTf] has been incorporated into polymer matrices — including poly(vinylidene fluoride), polyacrylonitrile, and poly(ethylene oxide) — to produce flexible gel polymer electrolytes for solid-state electrochemical devices, including flexible supercapacitors, solid-state batteries, and electrochromic devices.
- Cellulose and biomass dissolution: Imidazolium ionic liquids including [EMIM][OTf] demonstrate capacity to dissolve cellulose and lignocellulosic biomass, opening pathways for processing these renewable feedstocks into value-added products including biofuels, specialty fibers, and chemical building blocks under mild conditions without the harsh acid or base treatments required by conventional pulping processes.
Safety, Handling, and Environmental Considerations
While [EMIM][OTf] offers significant safety advantages over volatile organic solvents in terms of fire hazard and inhalation exposure, its environmental and toxicological profile warrants careful consideration. The compound is not acutely toxic by standard classifications, but imidazolium ionic liquids as a class have demonstrated ecotoxicological activity against aquatic organisms at elevated concentrations, with toxicity generally increasing with cation alkyl chain length — the ethyl group of [EMIM] places it in the lower-toxicity range of the imidazolium series. The fluorine-containing triflate anion is chemically stable and resistant to biodegradation, raising long-term environmental persistence concerns if the compound enters aquatic systems through improper disposal.
Recommended handling precautions include standard laboratory PPE — nitrile gloves, safety glasses, and laboratory coat — with particular attention to minimizing skin contact due to the potential for dermal absorption. Disposal should follow institutional chemical waste management protocols; the compound should not be poured down the drain due to its aquatic ecotoxicity and persistence. Storage in sealed containers away from strong oxidizing agents, strong bases, and moisture is advised. Despite these considerations, [EMIM][OTf]'s overall environmental risk profile compares favorably to many conventional solvents, particularly halogenated solvents, whose volatility, carcinogenicity, and persistence present more severe environmental and worker health risks under typical laboratory conditions.
Selecting [EMIM][OTf] for Your Application: Key Decision Criteria
[EMIM][OTf] is not a universal solution for every ionic liquid application, and informed selection requires matching its specific property profile against application requirements. It is the preferred choice when the following criteria apply:
- Low viscosity at room temperature is important — [EMIM][OTf] is among the less viscous common ionic liquids, making it preferable to longer-chain imidazolium triflates for mass-transport-dependent processes
- High ionic conductivity is required — its conductivity of ~8–9 mS/cm makes it one of the more conductive RTILs, suitable for electrochemical applications where minimizing internal resistance is critical
- Water miscibility is needed — unlike hydrophobic ionic liquids based on bis(trifluoromethylsulfonyl)imide (NTf₂) or hexafluorophosphate anions, [EMIM][OTf] is water-miscible, enabling aqueous biphasic systems and water-based processing steps
- Moderate electrochemical window is sufficient — where the ~4.1–4.3 V window of [EMIM][OTf] meets requirements without needing the wider windows achievable with NTf₂-based ionic liquids at the cost of lower conductivity
- Commercially available, well-characterized material is preferred — [EMIM][OTf] is widely available from specialty chemical suppliers in research and bulk quantities with comprehensive characterization data, reducing procurement and quality verification burden
As ionic liquid science continues to mature from academic curiosity to industrial implementation, [EMIM][OTf] occupies a well-established position as a benchmark material — extensively characterized, reliably synthesized, and sufficiently versatile to remain a first-choice consideration across electrochemistry, catalysis, and advanced materials processing for the foreseeable future.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体