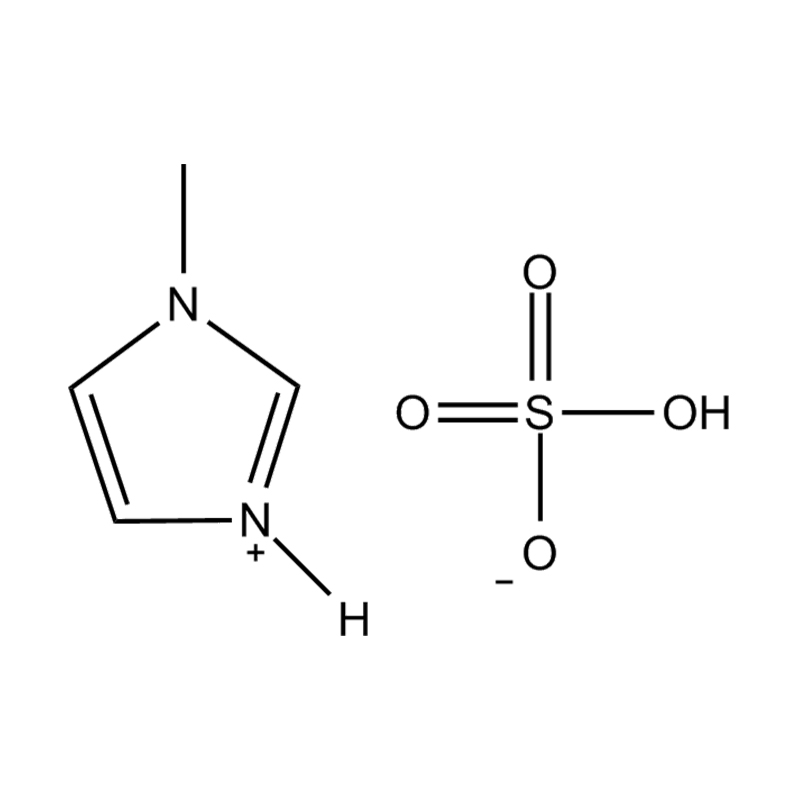
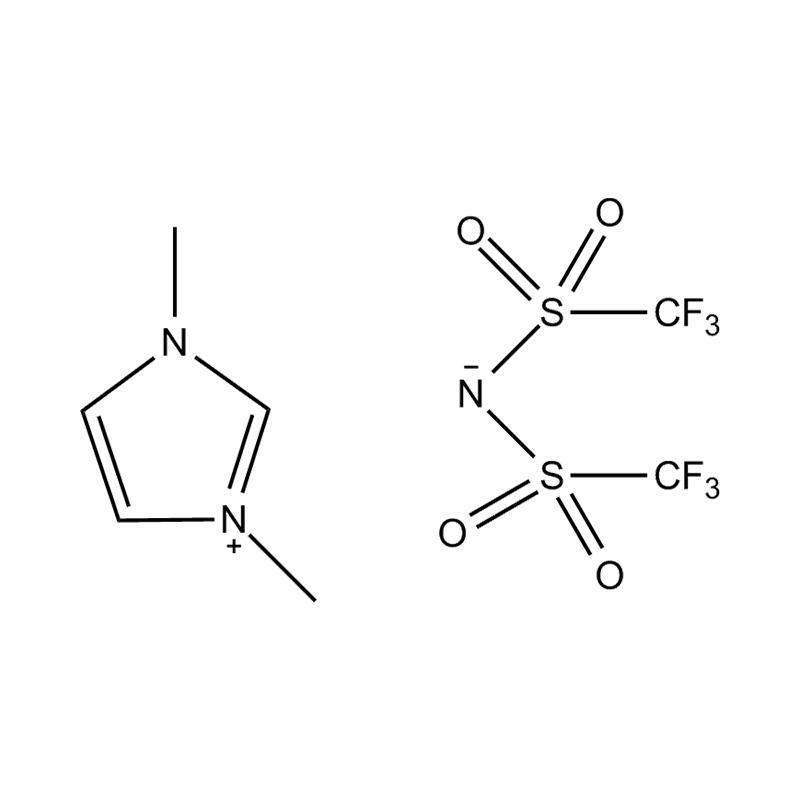
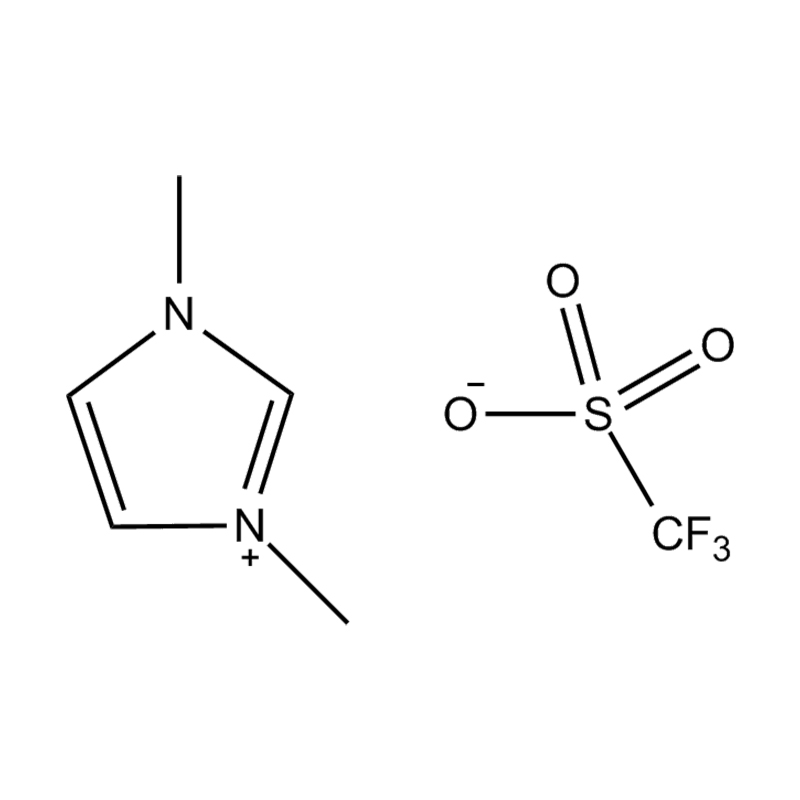
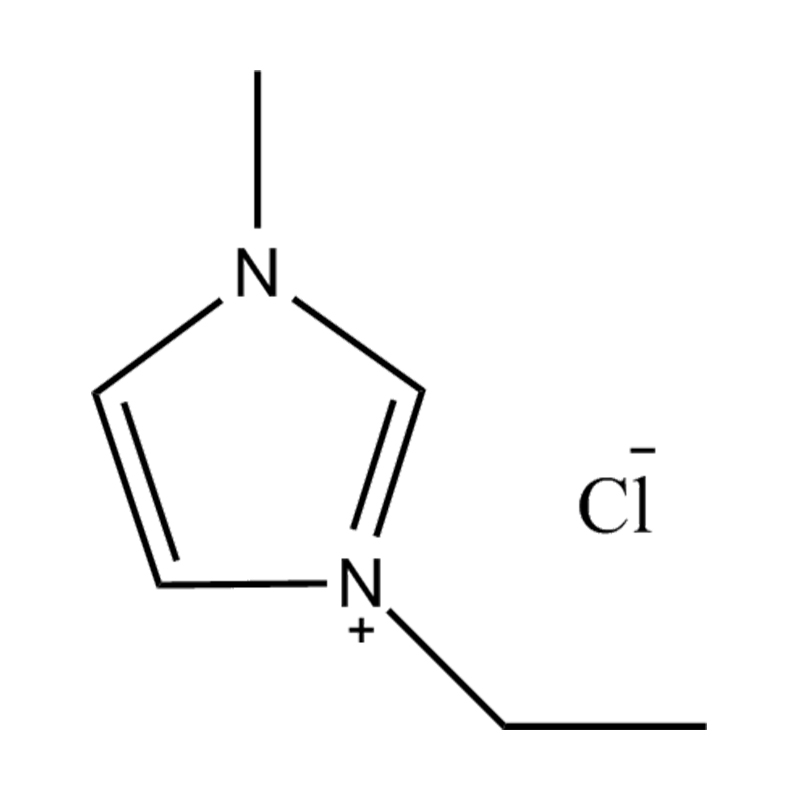
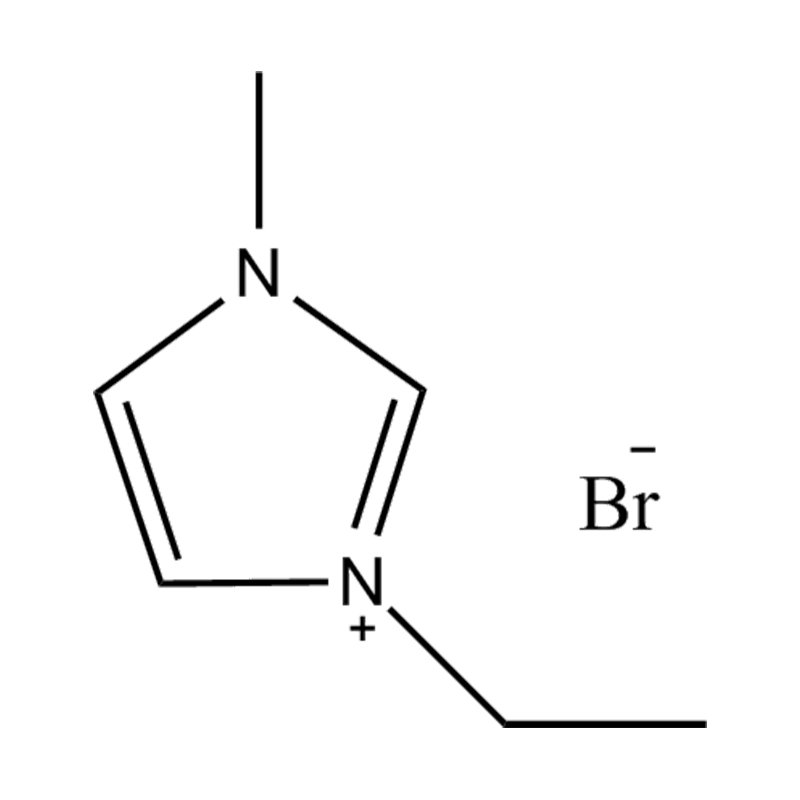
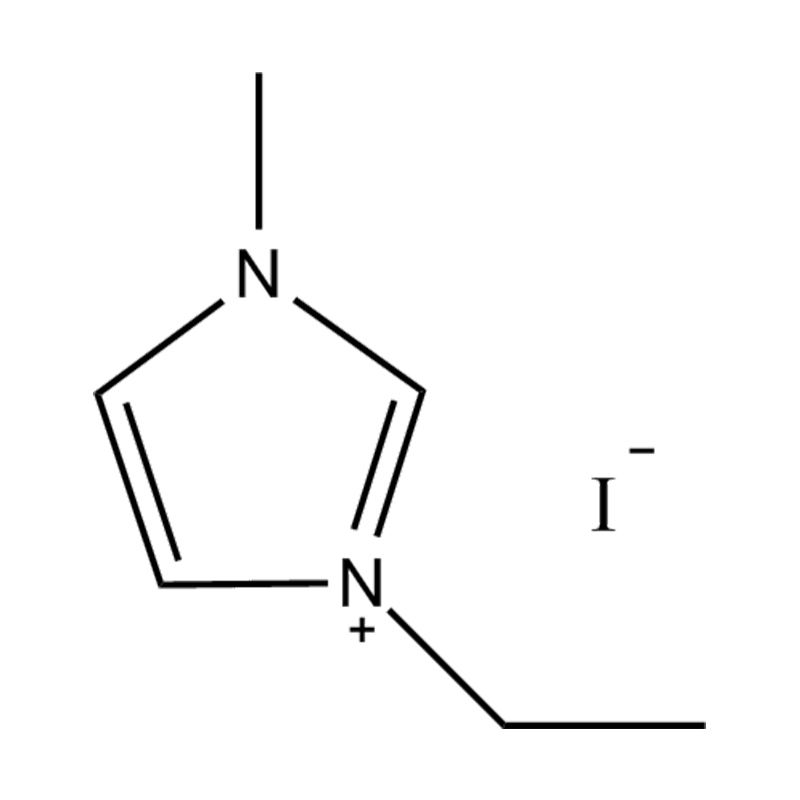
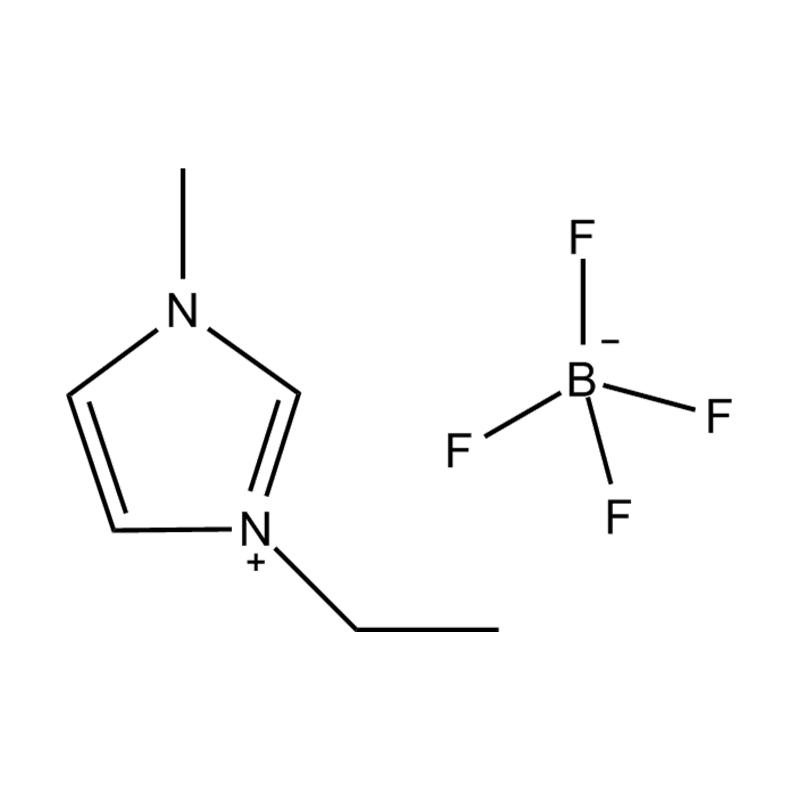
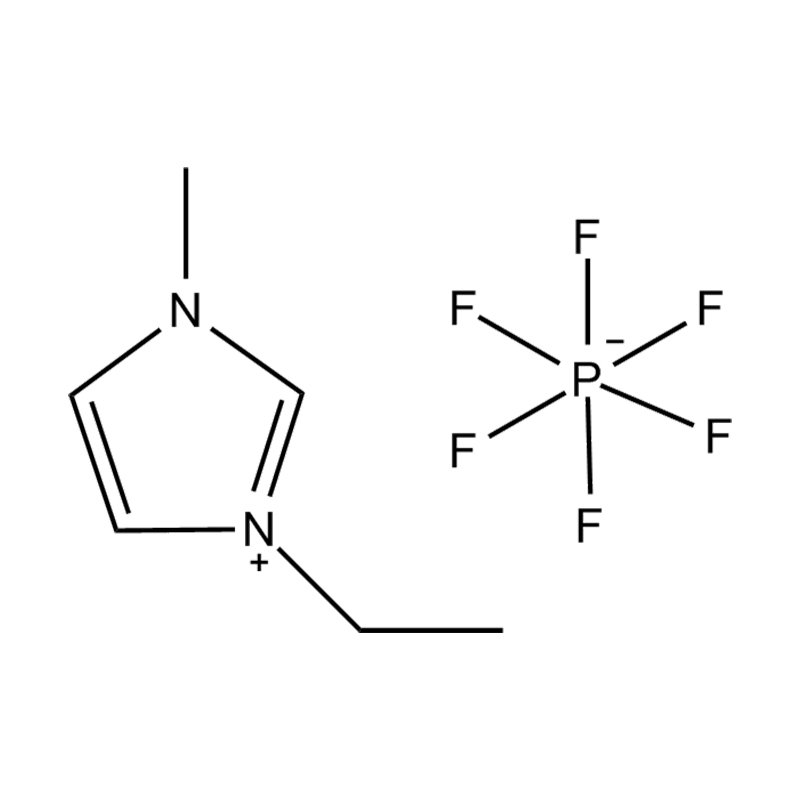
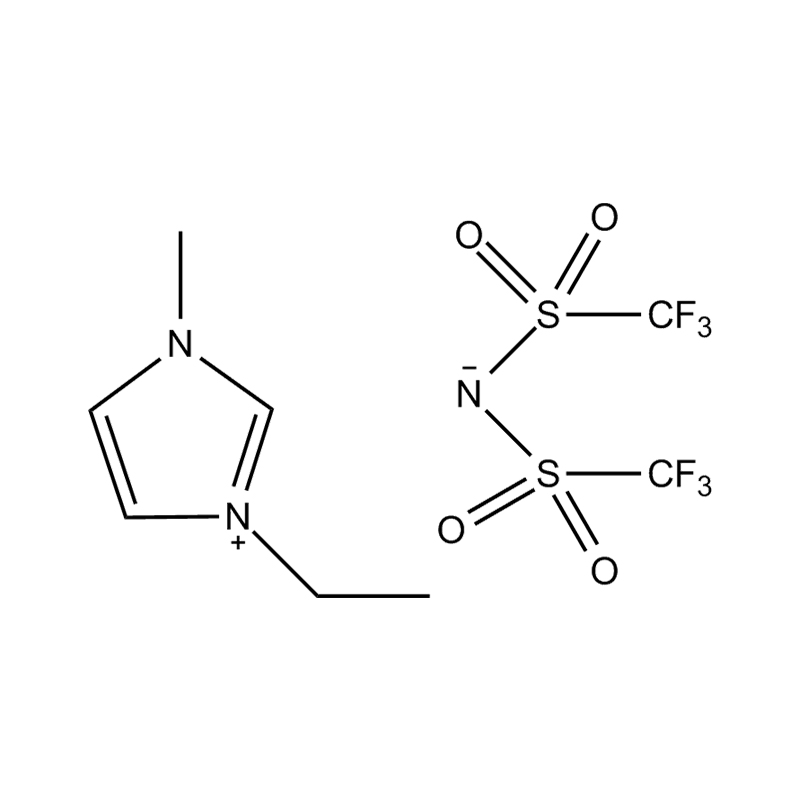
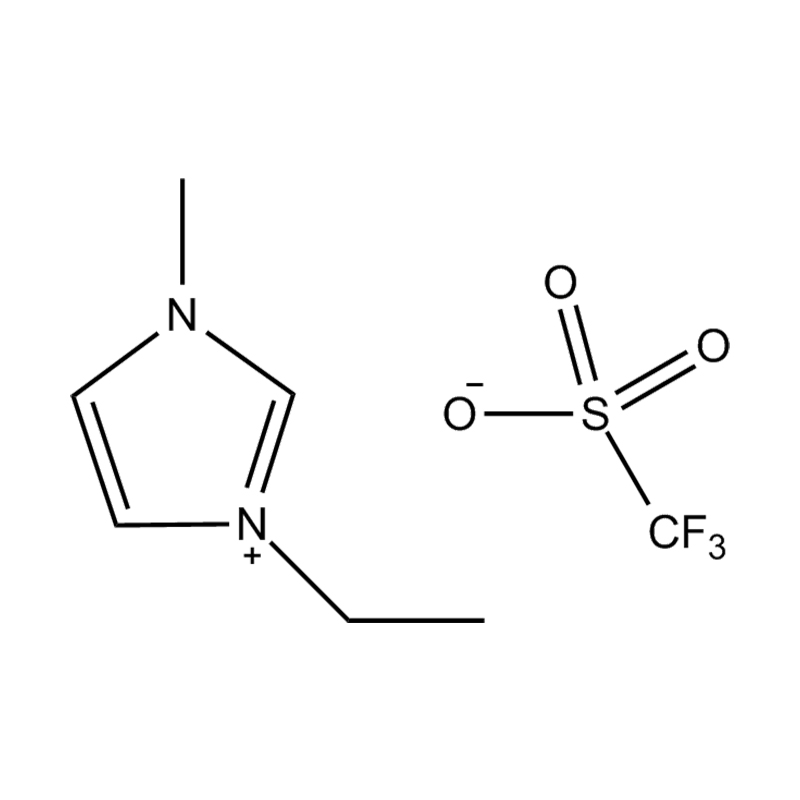
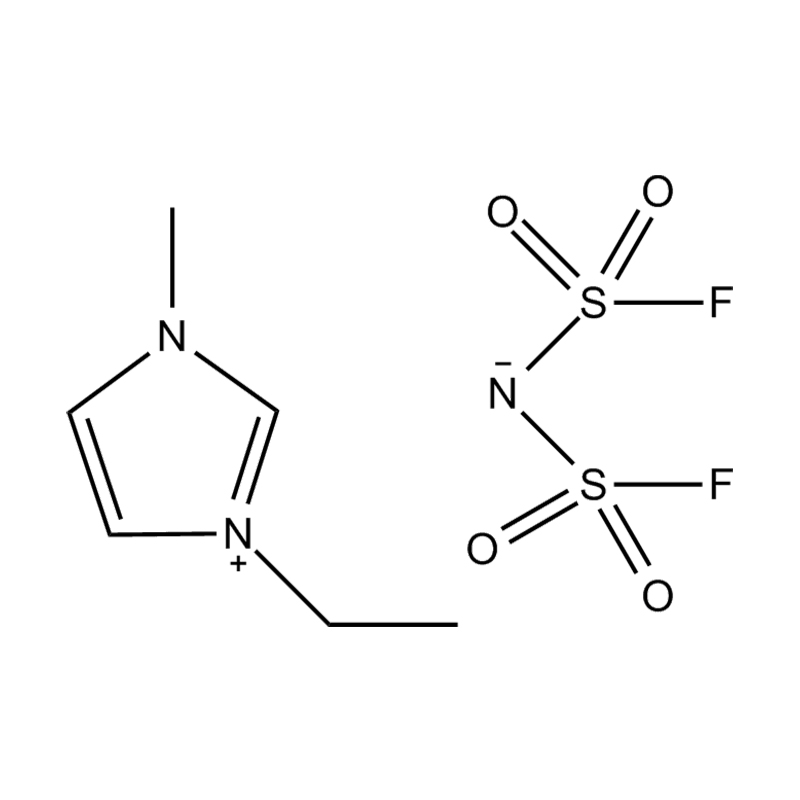
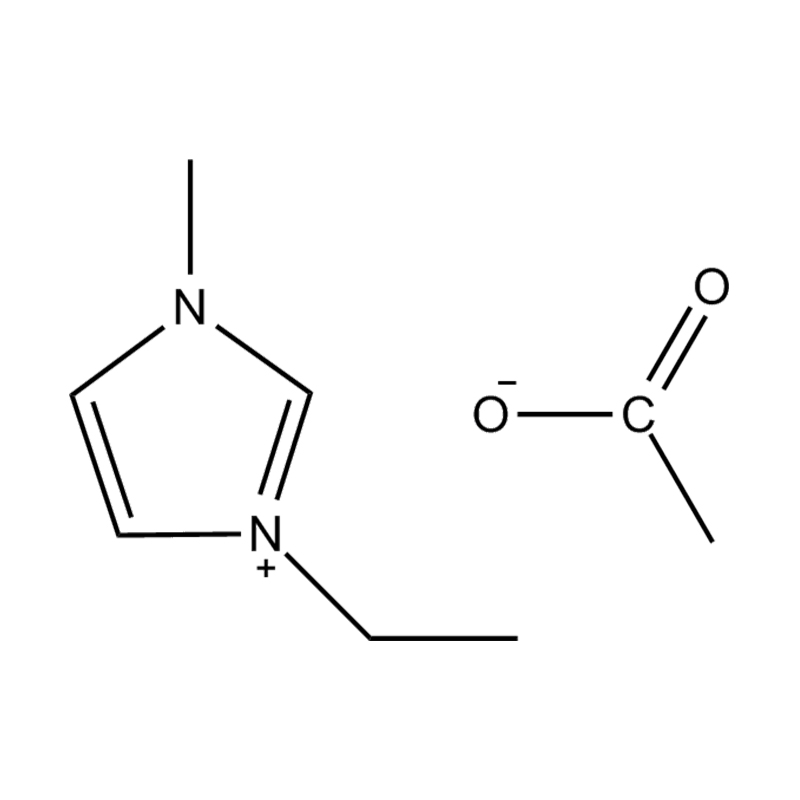
Using a catalyst in PET degradation offers several key advantages over traditional thermal and mechanical recycling methods. These benefits relate to efficiency, product quality, environmental impact, and resource recovery. Here's a detailed comparison:

1. Lower Energy Consumption
Catalytic Degradation:
Operates at significantly lower temperatures (often 180–250°C) compared to thermal methods.
Reduces energy input and operational costs.
Thermal Degradation:
Requires very high temperatures (above 400°C), making the process energy-intensive.
2. Selective Depolymerization
Catalysts enable controlled breakdown of PET into its original monomers—typically terephthalic acid (TPA) and ethylene glycol (EG) or bis(2-hydroxyethyl) terephthalate (BHET).
This chemical recycling allows recovery of high-purity monomers, which can be reused to make virgin-quality PET.
Mechanical Recycling, by contrast, typically only reprocesses PET into lower-grade plastics (downcycling), which limits reuse options.
3. Improved Recycling of Contaminated or Colored PET
Catalytic processes are less sensitive to impurities such as dyes, additives, and multilayer packaging.
Enables recycling of PET waste that is otherwise rejected in mechanical recycling streams.
4. Higher Product Value
The monomers recovered through catalysis can be reused in high-performance applications, including food-grade packaging.
In contrast, mechanically recycled PET (rPET) often suffers from color degradation, odor, and reduced mechanical strength.
5. Minimized Byproduct Formation
Well-designed catalysts promote specific reactions, reducing unwanted byproducts such as char, tars, or gases common in thermal degradation.
6. Faster Reaction Times
Catalysts accelerate depolymerization, allowing for shorter reaction times and higher throughput in industrial processes.
Thermal methods often require prolonged exposure to high heat, increasing operational wear and energy use.
7. Potential for Mild and Green Chemistry Conditions
Some catalysts (e.g., enzyme-based or ionic liquid catalysts) operate under mild conditions, potentially making the process more environmentally friendly and safer.
Summary Table
| Aspect | Catalytic Degradation | Thermal Degradation | Mechanical Recycling |
| Energy Requirement | Low to moderate | High | Low |
| Product Purity | High (monomers) | Low to moderate | Low (polymer quality drops) |
| Sensitivity to Contaminants | Low | Moderate to high | High |
| Environmental Impact | Lower | Higher (emissions, energy use) | Low to moderate |
| Scalability | Developing, but promising | Industrially proven | Widely used |
| End Product Value | High (virgin-grade possible) | Low to moderate | Low (downcycled products) |
Conclusion
Using a catalyst in PET degradation provides a pathway toward closed-loop recycling, where PET can be broken down and rebuilt without a significant loss in quality. This method stands out as a sustainable, efficient, and economically promising alternative to conventional thermal and mechanical recycling technologies.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体