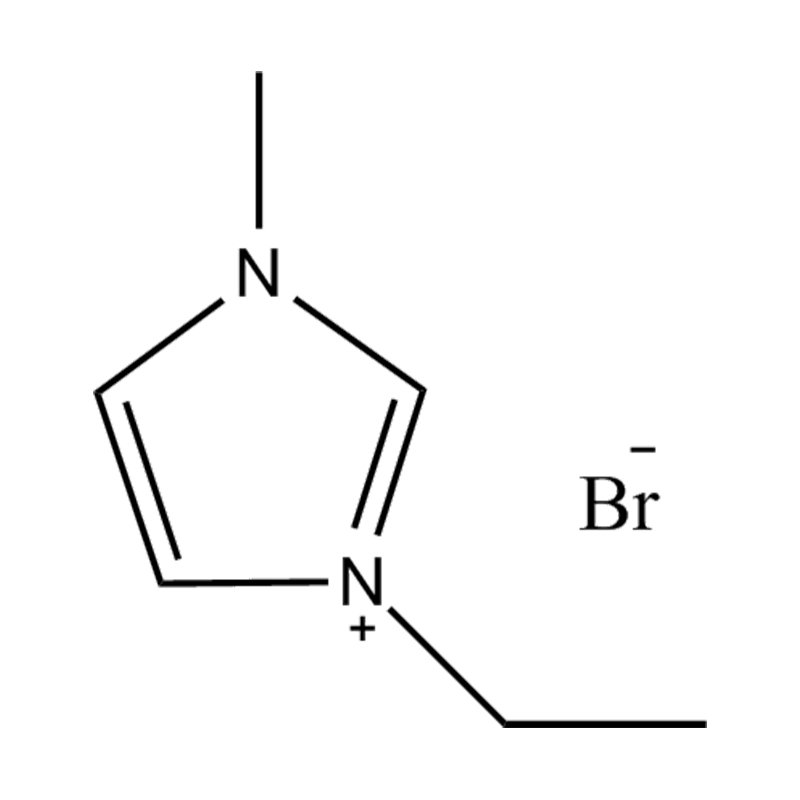
Ionic liquids (ILs) are salts composed entirely of ions that are liquid at or near room temperature — typically defined as melting below 100°C, with many remaining liquid well below 0°C. Unlike conventional molecular solvents, ionic liquids carry no net charge but consist entirely of a bulky organic cation paired with an inorganic or organic anion, a combination that disrupts the crystal lattice sufficiently to prevent solidification at ambient conditions. Their defining practical advantages — negligible vapor pressure, wide electrochemical window (typically 4–6 V), thermal stability to 200–400°C, and the ability to dissolve an exceptionally wide range of materials including cellulose, metal salts, and gases — make them uniquely valuable in industrial processes where conventional molecular solvents fail. More importantly, ionic liquids are inherently designable: by selecting from hundreds of available cations and anions, chemists can tune viscosity, polarity, miscibility, thermal range, and reactivity to meet precise process specifications — the foundation of the rapidly growing field of customized ionic liquids for industrial and specialty applications.

Content
- 1 What Makes Ionic Liquids Fundamentally Different from Conventional Solvents
- 2 The Architecture of Ionic Liquids: Cations and Anions That Define Performance
- 3 Industrial Ionic Liquids: Established Large-Scale Applications
- 4 Industrial Ionic Liquids in Practice: Scale, Cost, and Process Integration
- 5 Customized Ionic Liquids: Designing Molecular Properties for Specific Process Requirements
- 6 Specifying and Commissioning Customized Ionic Liquids: A Practical Framework
- 7 Environmental, Safety, and Regulatory Considerations for Ionic Liquids
What Makes Ionic Liquids Fundamentally Different from Conventional Solvents
Understanding why ionic liquids behave differently from water, alcohols, or organic solvents requires grasping their ionic nature. A conventional solvent like toluene or acetone consists of neutral molecules held together by weak van der Waals or dipolar forces — these molecules evaporate readily, creating vapor pressure, flammability hazard, and environmental exposure risk. An ionic liquid consists entirely of ions held together by strong electrostatic forces, but the size mismatch and asymmetry between the bulky organic cation and the anion prevent them from packing into a crystal lattice at ambient temperature.
This ionic structure produces a set of properties with no parallel in molecular solvents:
- Negligible vapor pressure: Most ionic liquids have vapor pressures below 10⁻¹⁰ Pa at 25°C — effectively zero for practical purposes. This eliminates evaporative loss, inhalation hazard, and VOC (volatile organic compound) emissions — a compliance advantage in jurisdictions with strict solvent emission regulations.
- Non-flammability: The absence of vapor pressure means most ionic liquids will not ignite or sustain combustion under normal handling conditions — a significant process safety advantage over conventional solvents in extraction, reaction, and electrochemical applications.
- Wide liquid temperature range: Many ionic liquids remain liquid from below −80°C to above 200°C — a temperature range no conventional solvent approaches without pressurization. This makes them useful as heat transfer fluids, high-temperature reaction media, and lubricants across extreme operating conditions.
- Wide electrochemical stability window: The absence of easily oxidized or reduced protons gives ionic liquids electrochemical windows of 4–6 V compared to approximately 1.2 V for water. This enables electrodeposition of metals (aluminum, titanium, rare earths) from ionic liquid baths that cannot be electrodeposited from aqueous solution.
- Excellent solvating power for diverse materials: The combination of ionic character, adjustable polarity, and dual donor-acceptor capability allows ionic liquids to dissolve materials that resist dissolution in both polar aqueous and non-polar organic solvents — including cellulose, lignin, chitin, metal oxides, and CO₂.
- Recyclability: Because ionic liquids are non-volatile, they can typically be recovered from reaction mixtures by evaporation of volatile co-solvents or products, then recycled — reducing ongoing solvent cost and waste disposal burden compared to conventional solvent processes.
The Architecture of Ionic Liquids: Cations and Anions That Define Performance
The specific properties of any ionic liquid are determined by the combination of its cation and anion. With hundreds of viable cation types and dozens of anion types commercially available or synthetically accessible, the theoretical number of distinct ionic liquid combinations exceeds 10¹⁸ — though in practice, a much smaller subset has been characterized and scaled for industrial use.
Common Cation Families
The cation is the larger, organic component and has the greatest influence on viscosity, density, and thermal stability. The four most industrially significant cation families are:
- Imidazolium ([Im]⁺): The most widely studied and commercially available cation family. 1-alkyl-3-methylimidazolium cations (e.g., [BMIM]⁺ — 1-butyl-3-methylimidazolium) combine moderate viscosity, good electrochemical stability, and excellent dissolving power. Their aromatic ring provides thermal stability to approximately 300°C. The main limitation is susceptibility to reaction under strongly basic conditions and moderate cost at large scale.
- Pyrrolidinium ([Pyrr]⁺): Saturated nitrogen heterocycle offering superior electrochemical stability (wider cathodic window than imidazolium) and better thermal stability to 350°C+. Preferred in battery and capacitor electrolyte applications where electrochemical stability is paramount. Typically higher viscosity than equivalent imidazolium ILs.
- Ammonium ([N]⁺) and Phosphonium ([P]⁺): Quaternary ammonium and phosphonium cations offer the highest thermal stability (phosphonium ILs stable to 400°C+) and are most relevant for high-temperature industrial process applications, lubrication, and heat transfer. Generally more viscous than imidazolium equivalents but lower-cost at scale using commercially available quaternary ammonium precursors.
- Pyridinium ([Py]⁺): Lower cost than imidazolium, useful in metal extraction, electroplating, and as reaction media for specific organic transformations. Less electrochemically stable than imidazolium and pyrrolidinium equivalents.
Anion Selection and Its Impact on Key Properties
The anion has the dominant influence on water miscibility, viscosity, and thermal decomposition behavior. Key industrially relevant anions include:
| Anion | Symbol | Water Miscibility | Viscosity Impact | Thermal Stability | Key Application |
|---|---|---|---|---|---|
| Bis(trifluoromethylsulfonyl)imide | [NTf₂]⁻ / [TFSI]⁻ | Immiscible | Low viscosity | Excellent (~400°C) | Batteries, electrochemistry, lubrication |
| Hexafluorophosphate | [PF₆]⁻ | Immiscible | Moderate | Good (~300°C) | Extraction, electrochemistry |
| Tetrafluoroborate | [BF₄]⁻ | Miscible | Low–Moderate | Good (~300°C) | Capacitors, reactions |
| Chloride | [Cl]⁻ | Miscible | High viscosity | Moderate | Cellulose dissolution, metal processing |
| Acetate | [OAc]⁻ | Miscible | High viscosity | Moderate (~200°C) | Biomass processing, biocatalysis |
| Dicyanamide | [N(CN)₂]⁻ | Miscible | Very low viscosity | Moderate (~250°C) | Dye-sensitized solar cells, reactions |
| Trifluoromethanesulfonate | [OTf]⁻ / [TfO]⁻ | Miscible | Moderate | Good (~350°C) | Catalysis, electrochemistry |
Industrial Ionic Liquids: Established Large-Scale Applications
Industrial ionic liquids have moved well beyond laboratory curiosity into deployed commercial processes across multiple sectors. The following applications represent areas where the technical performance advantages of ionic liquids have been demonstrated at production scale and where industrial adoption is growing.
BASF BASIL Process: The First Large-Scale Industrial IL Application
The BASIL (Biphasic Acid Scavenging utilizing Ionic Liquids) process, commercialized by BASF in 2002 for the production of alkoxyphenylphosphines (photoinitiator precursors), remains the most cited proof of ionic liquid industrial viability. In the conventional process, triethylamine was used as an acid scavenger, producing triethylammonium chloride — a solid that clogged equipment and severely limited reaction throughput. By replacing triethylamine with 1-methylimidazole, the reaction produces [HMIM]Cl (1-hexylmethylimidazolium chloride) — an ionic liquid that forms a separate liquid phase that can be continuously separated from the product. The result was a 10,000-fold increase in volumetric productivity, from 8 kg/m³·h to 690,000 kg/m³·h — demonstrating unambiguously that ionic liquids are not merely interesting chemistry but can enable transformative process engineering improvements.
Cellulose and Biomass Processing
One of the most technically significant industrial ionic liquid applications is the dissolution and processing of cellulose — a polymer that is insoluble in water and most organic solvents due to its extensive hydrogen bonding network. Ionic liquids with strongly hydrogen bond-accepting anions (chloride, acetate, phosphate) can dissolve cellulose at concentrations of up to 25% by weight at moderate temperatures (80–120°C), enabling homogeneous derivatization, fiber spinning, and composite formation processes impossible in conventional solvents.
The Ioncell process (developed at Aalto University, Finland) uses ionic liquids to produce regenerated cellulose fibers as a sustainable alternative to viscose rayon — without the toxic carbon disulfide and heavy metal sulfate effluents that make conventional viscose production environmentally problematic. The ionic liquid is designed for near-complete recovery (greater than 99.5% recycling efficiency) in a closed-loop process, addressing the economic concern about IL cost at scale.
Metal Extraction and Refining
Ionic liquids are increasingly important in hydrometallurgy and metal extraction, particularly for:
- Rare earth element (REE) separation: Conventional REE separation uses liquid-liquid extraction with organic solvents (kerosene, tributyl phosphate) and produces large volumes of radioactive aqueous waste. Ionic liquid extractants — particularly phosphonium and ammonium ILs — can achieve REE separation factors exceeding those of conventional systems with dramatically reduced environmental impact. The development of task-specific ionic liquids bearing chelating functional groups has further improved selectivity for individual REE elements.
- Electrodeposition of active metals: Aluminum, titanium, tantalum, and rare earth metals cannot be electrodeposited from aqueous solutions because water is reduced before the metal ion at the required cathodic potential. Ionic liquid electrolytes with their wide electrochemical window allow electrodeposition of these metals at atmospheric pressure and moderate temperature — enabling potential processes for coating, refining, and recycling of light metals and rare earths outside of high-temperature molten salt systems.
- Lithium-ion battery recycling: Ionic liquids can selectively dissolve and separate cathode active materials (LiCoO₂, LiNiMnCoO₂) from spent lithium-ion batteries, enabling recovery of lithium, cobalt, nickel, and manganese at purities suitable for reuse in battery manufacturing — a critical capability as battery recycling becomes mandatory under European and US regulatory frameworks.
Gas Capture and Separation
Ionic liquids show high and selective absorption of CO₂, SO₂, and H₂S from gas mixtures, making them candidates for post-combustion carbon capture and flue gas desulfurization. The physical absorption of CO₂ in [BMIM][PF₆] was one of the first ionic liquid gas capture applications studied, with solubility of approximately 0.6 mol CO₂ per mol IL at 40 bar and 25°C. More recently, task-specific ionic liquids bearing amine or azolide functional groups on the cation or anion achieve chemical absorption of CO₂ at near-ambient pressure, with absorption capacities of 0.5–1.0 mol CO₂ per mol IL at atmospheric pressure — comparable to aqueous amine scrubbing without the volatility and thermal degradation problems of amine solutions.
Lubrication and Tribology
Ionic liquids as lubricants and lubricant additives represent one of the fastest-growing industrial applications. Their key advantages in tribology are:
- Wide liquid temperature range — suitable as base lubricants from −60°C to 300°C+ without multi-grade additives
- Non-flammability — critical safety advantage in high-temperature machining, aerospace, and nuclear applications
- Electrostatic attraction of ionic liquid molecules to charged metal surfaces, forming an ordered boundary lubrication layer that reduces friction even at extreme pressure and low speed (stick-slip conditions)
- Demonstrated friction reduction of 30–60% and wear reduction of 70–90% compared to conventional mineral oil lubricants in steel-on-steel tribological testing
Phosphonium and imidazolium ILs with [NTf₂]⁻ or [PF₆]⁻ anions are the most commercially relevant lubricant ionic liquids, used neat or as 0.5–5% additives in conventional mineral or synthetic lubricant base stocks. The main limitation for steel applications has been corrosion — halide-containing ILs (particularly chloride anion) cause steel corrosion at tribological contact points, driving development of halide-free IL lubricants for ferrous applications.
Electrochemical Energy Storage
In electrochemical double-layer capacitors (EDLCs or supercapacitors) and next-generation lithium-based batteries, ionic liquid electrolytes address the two main failure modes of conventional carbonate-based electrolytes: flammability (eliminated by ionic liquid non-flammability) and electrochemical window limitations (the 1.2 V window of aqueous electrolytes and 4.5 V window of organic electrolytes versus 5–6 V for ILs). Pyrrolidinium [NTf₂]⁻ ionic liquids are the most advanced in battery applications, with commercial ionic liquid electrolytes used in specialty high-temperature lithium-ion cells and in lithium-sulfur battery development where the ionic liquid suppresses polysulfide shuttle reactions that cause conventional cell capacity fade.
Industrial Ionic Liquids in Practice: Scale, Cost, and Process Integration
The transition from laboratory to industrial scale introduces challenges specific to ionic liquids that do not arise with conventional solvents. Understanding these challenges is essential for process engineers evaluating ionic liquid integration.
Cost at Scale
The cost of ionic liquids at commodity scale depends heavily on the cation and anion selected. Simple imidazolium chloride or bromide ILs can be produced at $5–50/kg at multi-tonne scale using commercially available methylimidazole and alkyl halide precursors. [NTf₂]⁻-containing ILs cost more due to the fluorinated anion (lithium bistriflimide salt is a precursor at $30–60/kg), typically resulting in finished IL costs of $50–200/kg at scale. Highly functionalized or task-specific ionic liquids for specialty applications remain at $500–5,000/kg or above at current production volumes.
The economic case for industrial ionic liquids therefore depends critically on recyclability. A process that recovers and reuses ionic liquid at 99% efficiency per cycle amortizes the initial IL cost across thousands of batches — the effective IL cost per unit product becomes comparable to conventional solvents. Processes where ionic liquid recovery falls below 95% per cycle face ongoing IL replacement costs that undermine the economic case at current IL pricing.
Viscosity: The Primary Process Engineering Challenge
Ionic liquids are significantly more viscous than water or common organic solvents at ambient temperature. Typical viscosities at 25°C range from 20 mPa·s (low-viscosity ILs such as [EMIM][N(CN)₂]) to over 1,000 mPa·s (high-viscosity ILs such as [BMIM][Cl]), compared to 1 mPa·s for water and 0.6 mPa·s for acetone. High viscosity reduces mass transfer rates, increases pumping energy requirements, and limits diffusion-controlled reaction rates. Process engineers typically address viscosity through:
- Temperature elevation: Ionic liquid viscosity decreases sharply with temperature — a 20°C increase typically reduces viscosity by 50–70%. Operating at 60–80°C rather than ambient temperature is often sufficient to achieve acceptable mass transfer characteristics.
- Co-solvent addition: Small additions of water (5–15% by mass) or low-viscosity organic co-solvents can reduce viscosity by one to two orders of magnitude while preserving most of the IL's beneficial properties, depending on application requirements.
- Anion selection: Choosing low-viscosity anions ([NTf₂]⁻, [N(CN)₂]⁻, [OTf]⁻) at the design stage is the most effective way to minimize viscosity while maintaining other target properties.
Water Sensitivity and Purity Requirements
Many ionic liquids — particularly those with water-miscible anions (halides, acetate, tetrafluoroborate) — are hygroscopic and absorb atmospheric moisture, which changes their properties significantly: density decreases, viscosity drops, and electrochemical window narrows as water is incorporated. Industrial processes using moisture-sensitive ionic liquids require inert atmosphere handling and storage, or deliberate water management to maintain target IL composition within specification. This adds process complexity compared to conventional organic solvents but is manageable with appropriate engineering controls.
Customized Ionic Liquids: Designing Molecular Properties for Specific Process Requirements
The most powerful — and most commercially significant — aspect of ionic liquid technology is the ability to design custom ILs with properties targeted to a specific application. Unlike conventional solvents, where the engineer selects from a fixed catalogue of commercially available options, ionic liquids can be engineered at the molecular level to optimize any combination of properties. This is the basis of the growing market for customized ionic liquids tailored for specific industrial, pharmaceutical, agricultural, and specialty chemical applications.
The Customization Framework: What Can Be Tuned and How
Ionic liquid customization operates at three levels: cation modification, anion selection, and functional group incorporation.
- Alkyl chain length on the cation: Increasing the alkyl chain length on the imidazolium, pyrrolidinium, or ammonium cation increases hydrophobicity, decreases water miscibility, increases viscosity, and — beyond chain lengths of approximately C₈ — introduces amphiphilic character (self-assembly into micellar structures in water). For example, moving from [EMIM]⁺ (ethyl) to [OMIM]⁺ (octyl) with the same [NTf₂]⁻ anion shifts the IL from water-miscible to water-immiscible, enabling liquid-liquid biphasic extraction designs.
- Functional group incorporation (task-specific ionic liquids): Chemical functionality can be attached directly to the cation or anion to give the IL a specific chemical interaction capability beyond its solvent properties. Examples include: amine-functionalized cations for CO₂ chemisorption; thiol or thioether groups for selective mercury extraction; crown ether pendants for alkali metal ion extraction; chiral centers for asymmetric catalysis and chiral resolution; polymerizable groups for IL-based solid electrolytes and membranes.
- Protic vs. aprotic design: Protic ionic liquids (PILs) are formed by proton transfer from a Brønsted acid to a Brønsted base, producing an IL in which the cation retains a transferable proton (e.g., [EtNH₃][NO₃]). PILs are distinct from aprotic ILs in their ability to participate in proton-transfer reactions and hydrogen bonding — making them relevant as fuel cell electrolytes, proton-conducting membranes, and media for acid-catalysed organic reactions.
- Deep eutectic solvents (DES) — a customization extension: Deep eutectic solvents are mixtures of hydrogen bond donors and acceptors (e.g., choline chloride with urea, glycerol, or malonic acid) that form liquid eutectic mixtures with properties similar to ionic liquids but at significantly lower cost and using entirely bio-derived components. DES represent the low-cost, high-volume extension of the customizable IL concept and are attracting substantial industrial interest for metal processing, pharmaceutical extraction, and electrochemistry applications.
Computational Design of Customized Ionic Liquids
The experimental screening of ionic liquid property combinations across even a small subset of possible cation-anion pairs is prohibitively expensive at laboratory scale. Computational approaches — molecular dynamics (MD) simulation, quantum chemical calculations, and machine learning models trained on experimental IL property databases — are increasingly used to predict ionic liquid properties before synthesis, narrowing the design space to the most promising candidates for experimental validation.
Property prediction models are now available for viscosity, density, melting point, decomposition temperature, and CO₂ solubility for common cation-anion combinations, with prediction accuracy typically within 10–20% of experimental values for well-characterized IL families. The ILThermo database (NIST) and the IUPAC Ionic Liquids Database contain experimental data for over 30,000 IL-property combinations, providing the training data for increasingly accurate predictive models. For industrial clients commissioning custom IL synthesis, computational pre-screening reduces the number of synthesis-and-test cycles needed to reach the target specification, reducing development time and cost.
Pharmaceutical and Agricultural Active Ingredient Ionic Liquids
A particularly innovative category of customized ionic liquids combines an active pharmaceutical ingredient (API) or agricultural active as one of the ionic components. An API ionic liquid (API-IL) incorporates the drug molecule as the cation or anion, paired with a biocompatible counter-ion. This approach offers several pharmaceutical advantages:
- Improved bioavailability: Crystalline drug salts can have poor dissolution rates that limit oral bioavailability. Converting a drug to its ionic liquid form eliminates the crystalline state, giving amorphous material with faster dissolution and potentially higher absorption.
- Combination therapy in a single compound: A drug cation can be paired with a drug anion (two active ingredients as the two IL components), delivering both components in a fixed, equimolar ratio with a single compound.
- Transdermal delivery: The liquid nature and low melting point of API-ILs facilitates transdermal formulation — several API-IL patch formulations have demonstrated significantly improved skin permeation compared to conventional salt forms.
In agriculture, ionic liquid formulations of herbicides (e.g., ionic liquid forms of glyphosate, dicamba) have demonstrated improved plant uptake, reduced drift risk, and lower required application rates compared to conventional salt formulations — addressing both agronomic performance and environmental impact concerns simultaneously.
Specifying and Commissioning Customized Ionic Liquids: A Practical Framework
For industrial buyers and R&D departments seeking to commission customized ionic liquids from specialist suppliers, the specification and procurement process requires clarity on parameters that are unlike conventional chemical procurement.
Key Specification Parameters to Define
| Parameter | Why It Matters | Typical Specification Range |
|---|---|---|
| Halide impurity content | Residual halide (Cl⁻, Br⁻) from synthesis causes corrosion, catalyst poisoning, and altered electrochemistry | <10 ppm (high purity), <100 ppm (standard) |
| Water content | Changes viscosity, density, electrochemical window; must be defined for reproducible process performance | <50 ppm (anhydrous), <500 ppm (standard), or defined % water |
| Viscosity at operating temperature | Determines pump sizing, mass transfer rates, and mixing energy requirements | Specify at process temperature(s), ±10% |
| Thermal decomposition onset | Defines safe operating temperature ceiling for process design | T₅% (5% mass loss by TGA) as minimum threshold |
| Electrochemical window | Critical for electrolyte and electrodeposition applications; determines what reactions are possible without electrolyte decomposition | Specify cathodic and anodic limits vs. reference electrode |
| Color and appearance | Yellow/brown coloration indicates organic impurities from synthesis; relevant for optical and pharmaceutical applications | APHA color <50 (colorless to pale yellow) |
| Ionic purity (NMR) | Confirms correct cation:anion identity and stoichiometry; detects synthesis byproducts | ≥98% pure (standard), ≥99.5% (high purity) |
Synthesis Routes and Their Impact on Purity
The most common industrial synthesis routes for ionic liquids involve quaternization (alkylation of a nitrogen base with an alkyl halide to form a halide IL) followed by anion metathesis (replacing the halide anion with the target anion using a silver salt, Lewis acid, or ion exchange resin). The halide intermediate step is the primary source of residual halide contamination in the final IL — a critical quality issue for electrochemical and catalytic applications. Synthesis routes that avoid halide intermediates — for example, direct neutralization of Brønsted acid-base pairs for protic ILs, or reaction of tertiary amines with dialkyl sulfate esters — can produce halide-free ILs without the metathesis step, but are limited to specific IL structures.
Suppliers of customized ionic liquids should provide characterization data including ¹H NMR and ¹³C NMR spectra confirming structure, ion chromatography or potentiometric titration for halide content, Karl Fischer titration for water content, and thermogravimetric analysis (TGA) for thermal stability — as a minimum analytical package for process-grade custom ILs.
Environmental, Safety, and Regulatory Considerations for Ionic Liquids
The early marketing of ionic liquids as inherently "green" solvents due to their non-volatility requires significant nuance. While the negligible vapor pressure eliminates atmospheric VOC emissions — a genuine environmental advantage — several important environmental and safety considerations must be evaluated for any industrial ionic liquid application.
Aquatic Toxicity
Ecotoxicological studies have consistently demonstrated that imidazolium ionic liquids exhibit moderate to high aquatic toxicity — particularly toward algae, crustaceans, and bacteria — with EC₅₀ values for common ILs ranging from approximately 1 mg/L to 100 mg/L depending on alkyl chain length (longer chains = higher toxicity) and anion. This means that accidental aquatic release of ionic liquids must be prevented with equivalent rigor to conventional organic solvents, despite their non-volatility. Biodegradability of most commercially available ionic liquids is poor to moderate — fluorinated anion-containing ILs ([NTf₂]⁻, [PF₆]⁻) are particularly persistent in the environment. This drives interest in biodegradable IL design using natural acid or amino acid-derived anions and choline-based cations.
REACH and CLP Registration
Ionic liquids are subject to REACH registration requirements in the EU for volumes above 1 tonne per year per manufacturer or importer. Most commercially available ionic liquids used in research quantities have not been formally REACH-registered as industrial chemicals, meaning that scaling an IL-based process to industrial volumes requires either commissioning a formal registration (costly and time-consuming) or purchasing from a supplier who has already registered the specific IL. The absence of REACH data for many IL structures is a significant practical barrier to rapid industrial adoption that is often underappreciated during laboratory feasibility studies.
Designing for Reduced Environmental Impact
The design principles for more environmentally acceptable ionic liquids include:
- Choline-based cations: Choline is a naturally occurring quaternary ammonium compound (vitamin B4) with low toxicity and good biodegradability. Choline-based ILs — particularly choline chloride combined with hydrogen bond donors (forming deep eutectic solvents) — offer the most promising path to low-cost, low-toxicity, biodegradable ionic media.
- Amino acid anions: Replacing halide, fluorinated, or phosphate anions with amino acid anions (glycinate, alaninate, prolinate) produces ILs with substantially better biodegradability and lower aquatic toxicity, while maintaining useful solvent properties for some applications.
- Avoidance of persistent fluorinated anions: [NTf₂]⁻ and [PF₆]⁻ anions, while excellent for electrochemical and lubricant applications, contain fluorine that generates persistent fluoride and PFAS-related compounds upon degradation. For applications where these anions are not functionally required, their substitution with non-fluorinated alternatives ([OTf]⁻ excepted) is an environmental design improvement.


 English
English Deutsch
Deutsch Español
Español 中文简体
中文简体